

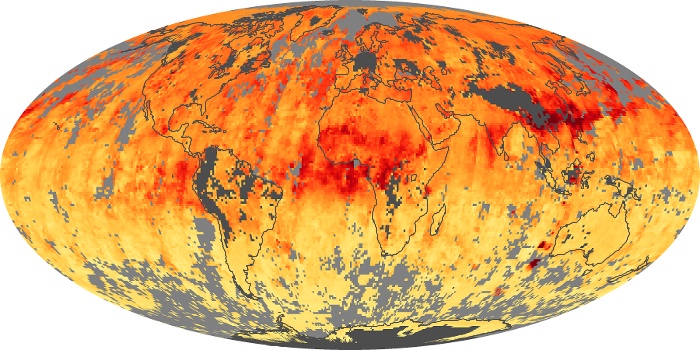
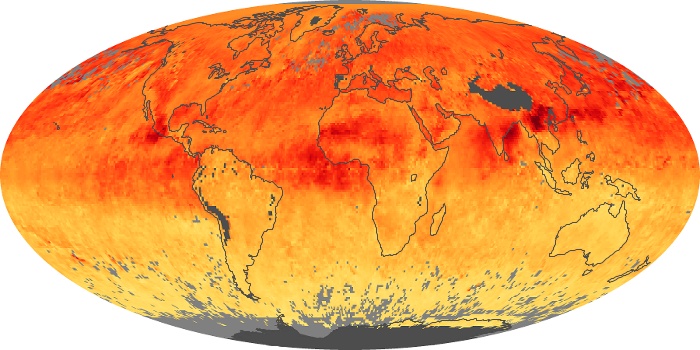
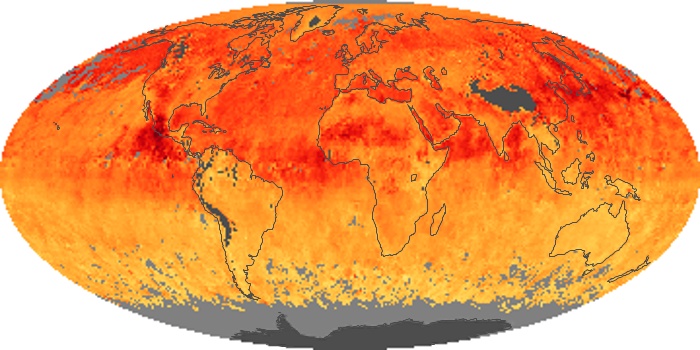
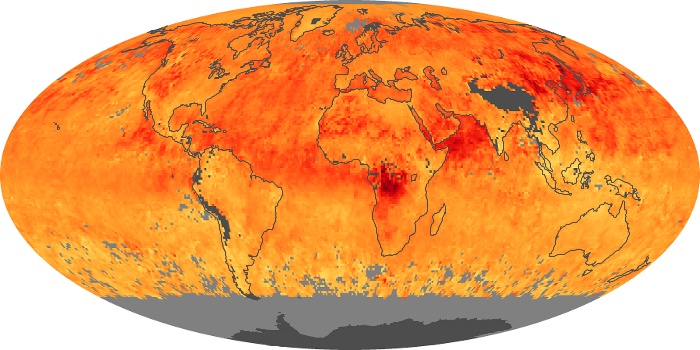
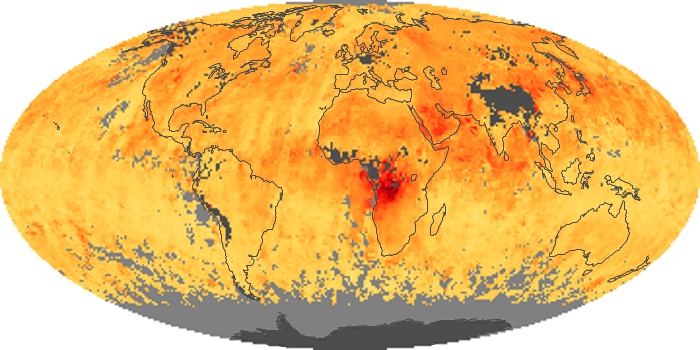
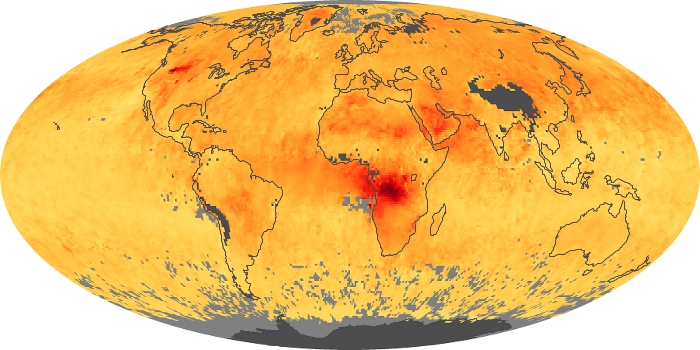
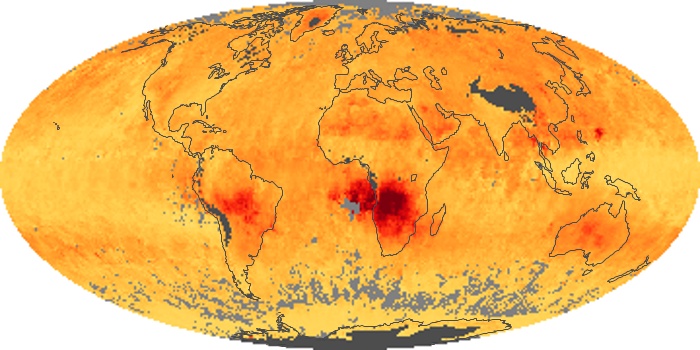
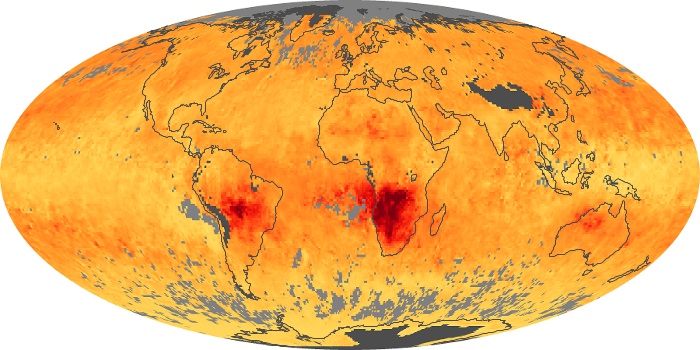
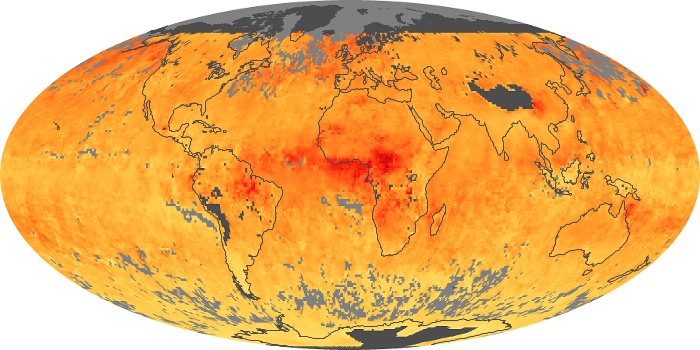
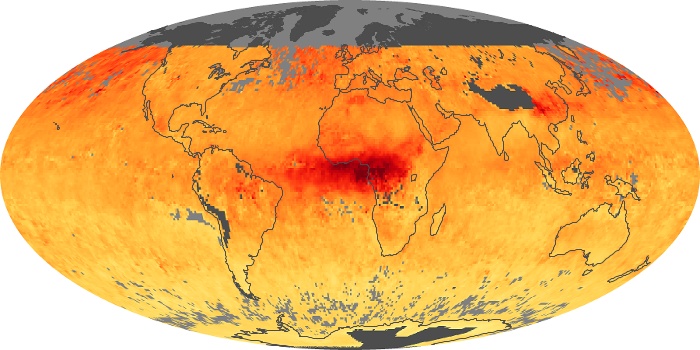
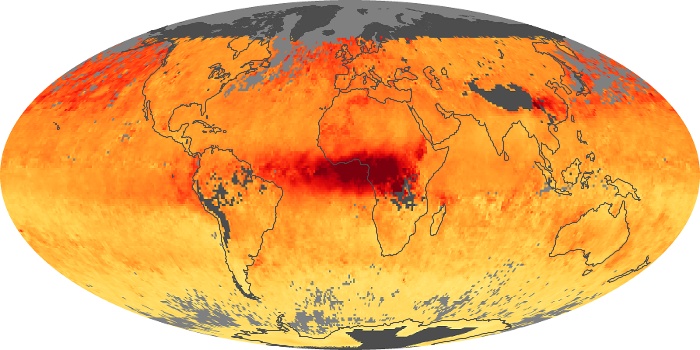
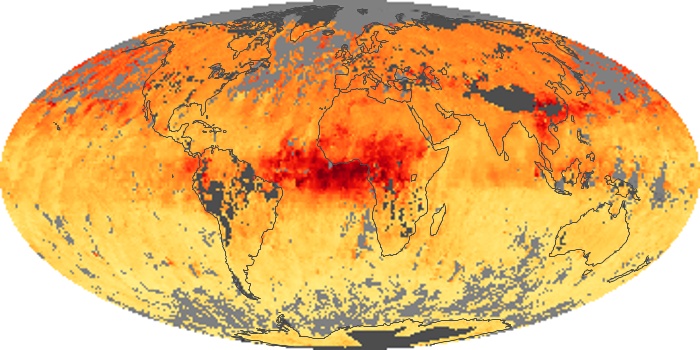
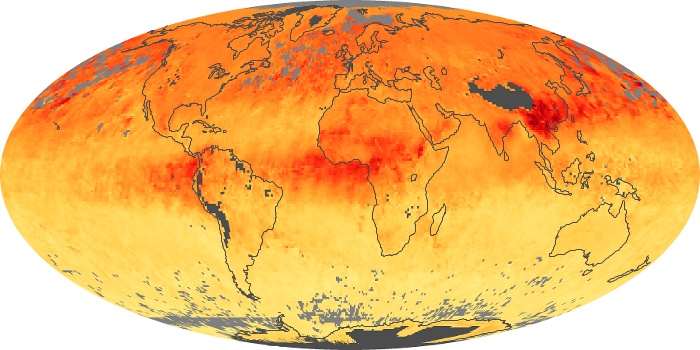
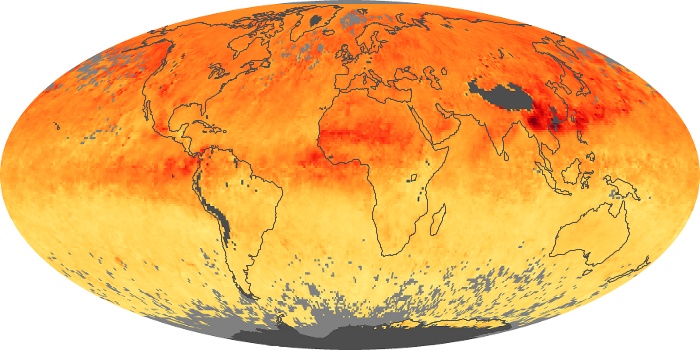



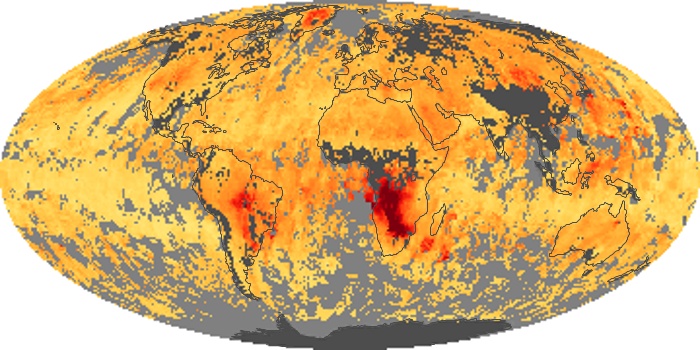
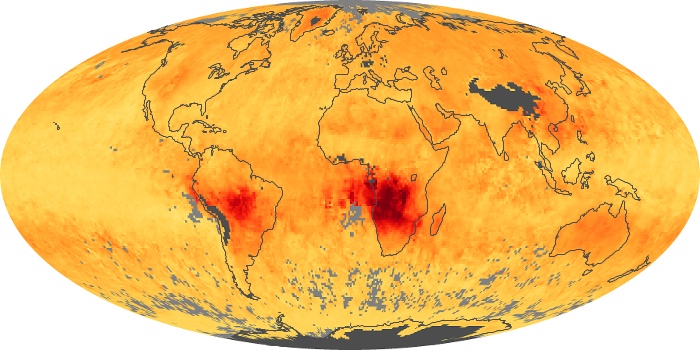
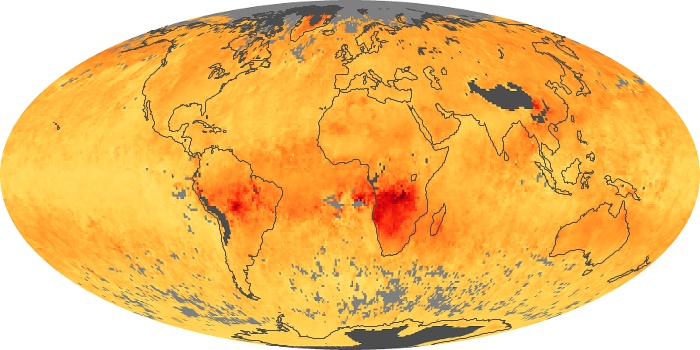
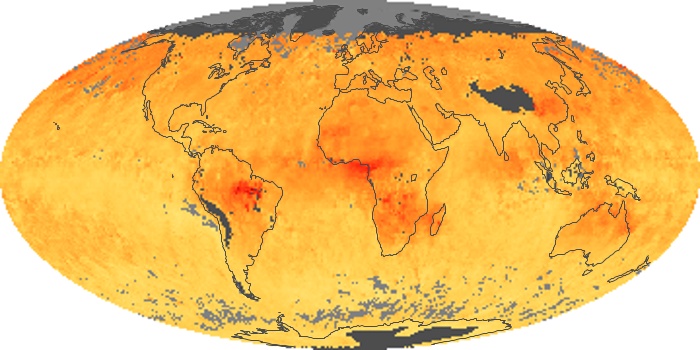
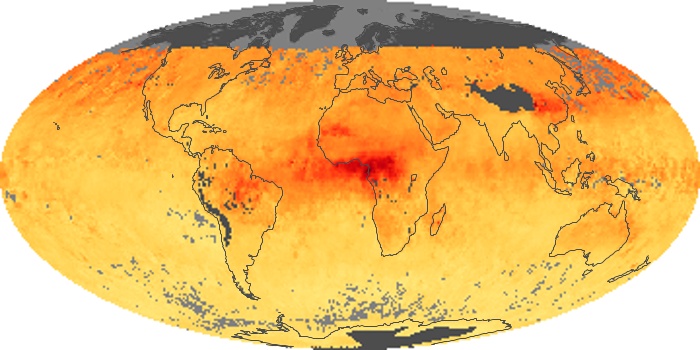
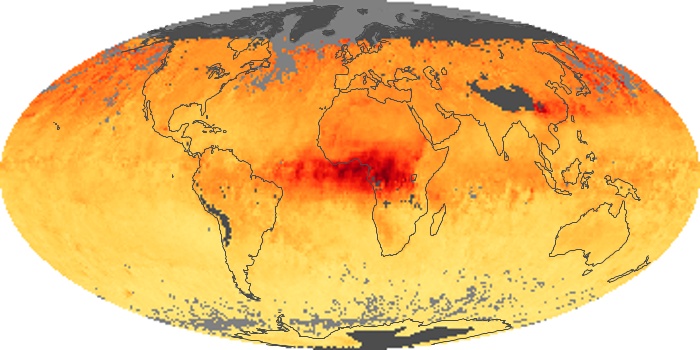
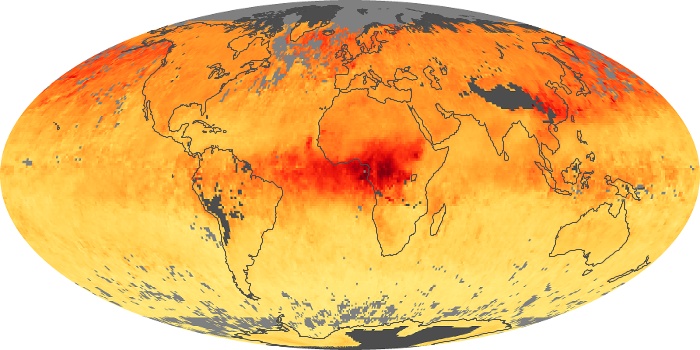
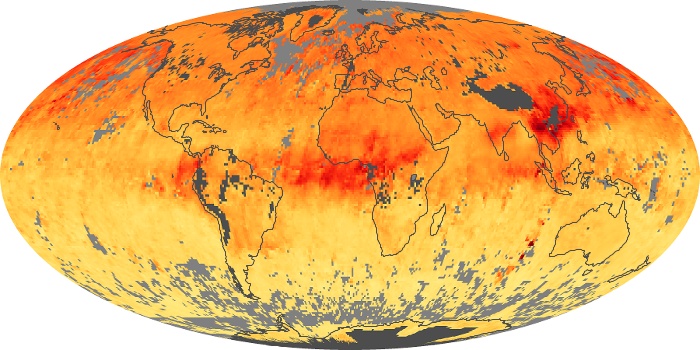
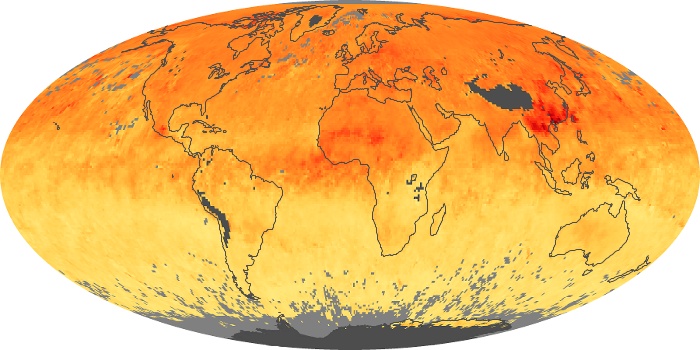
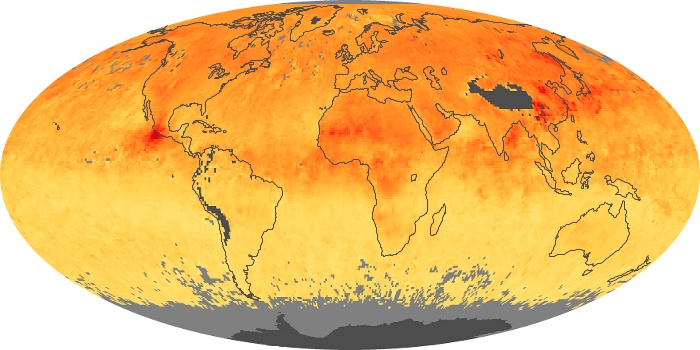
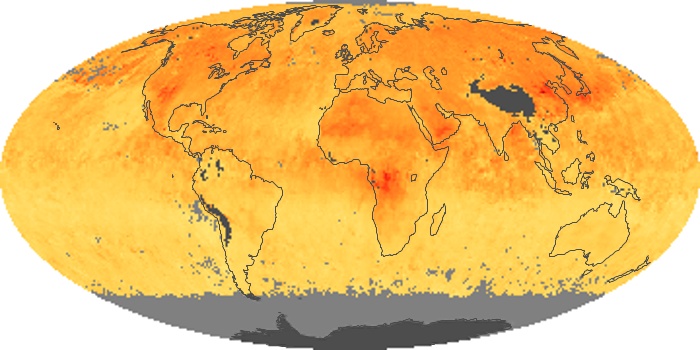
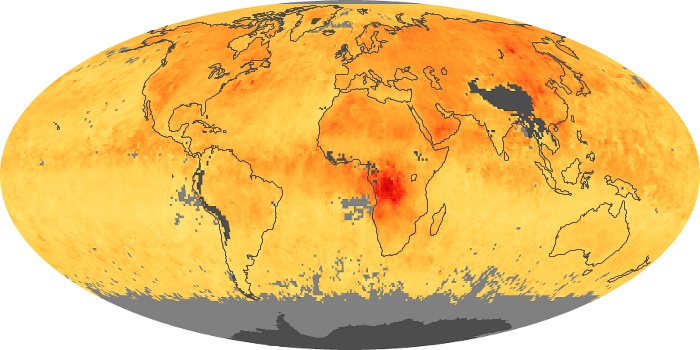
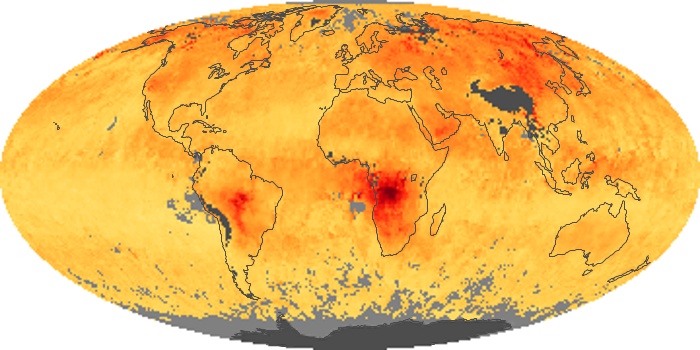
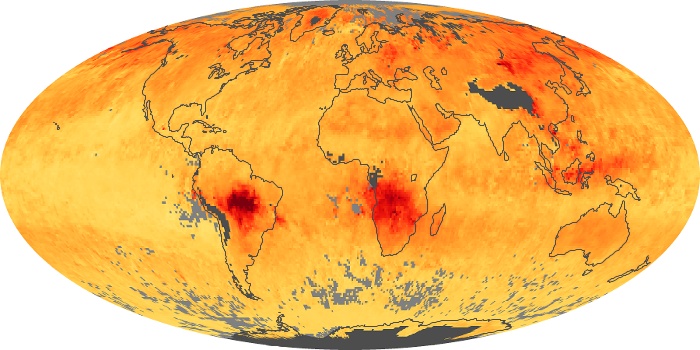
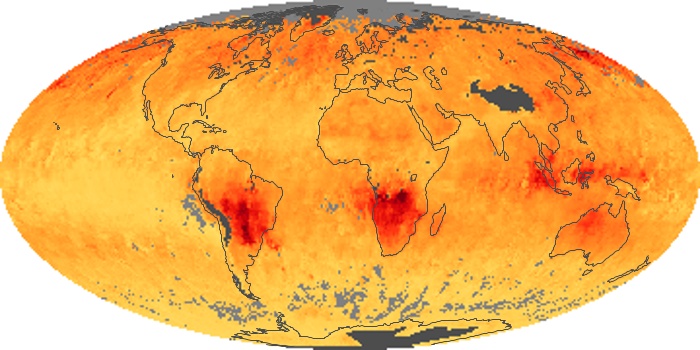
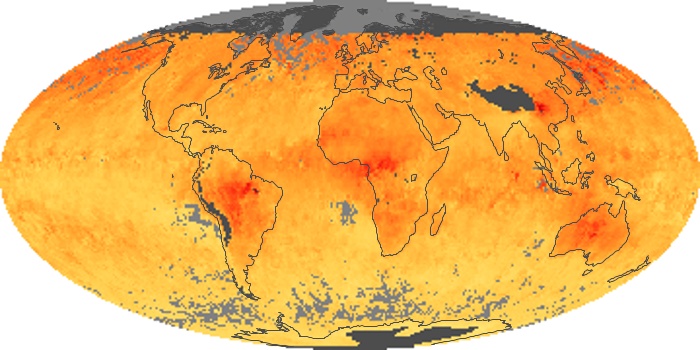
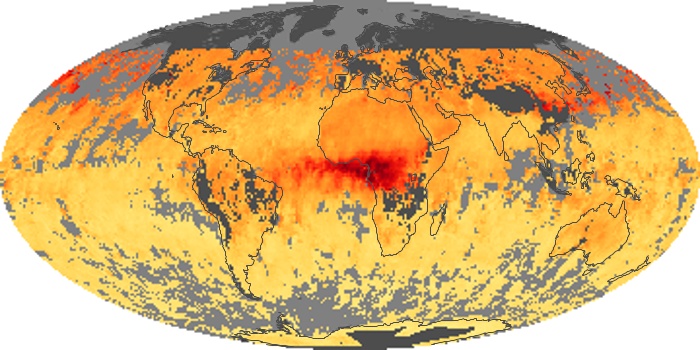
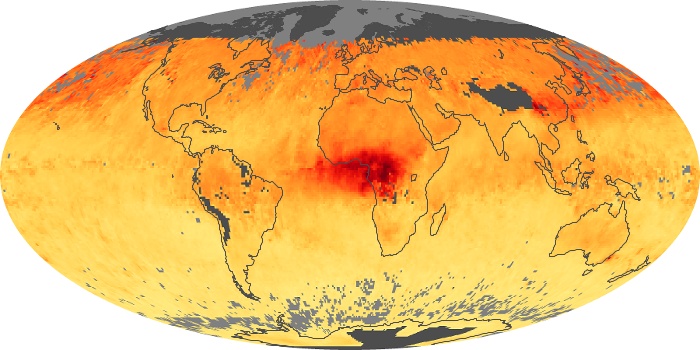
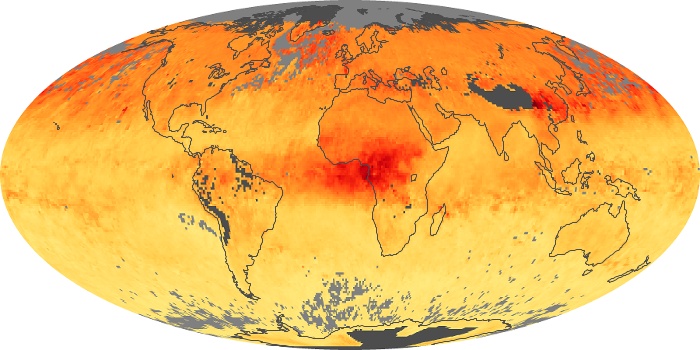
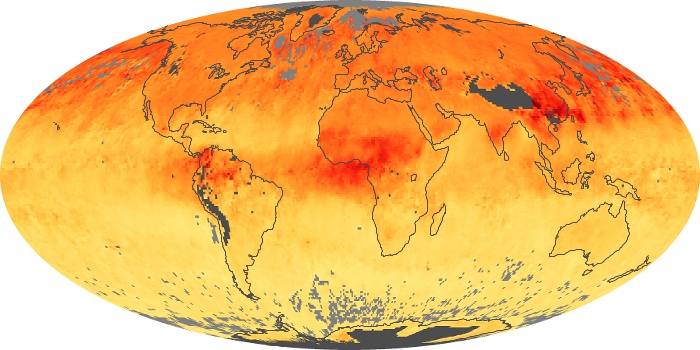
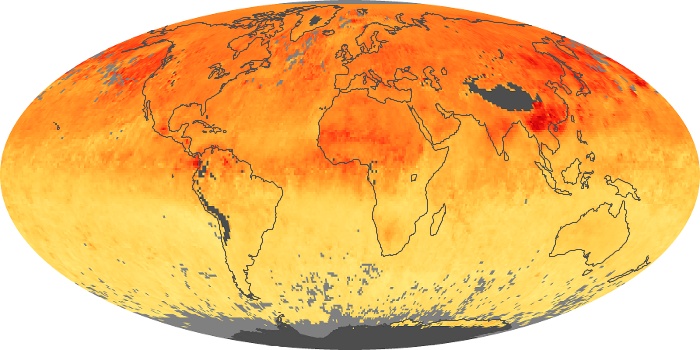
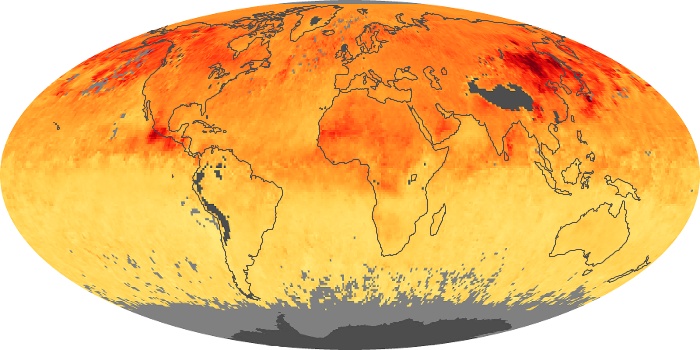
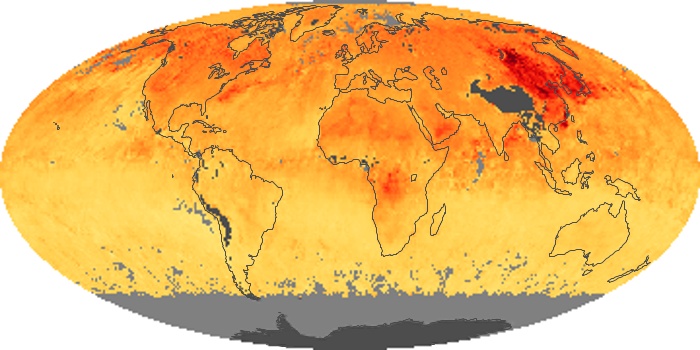
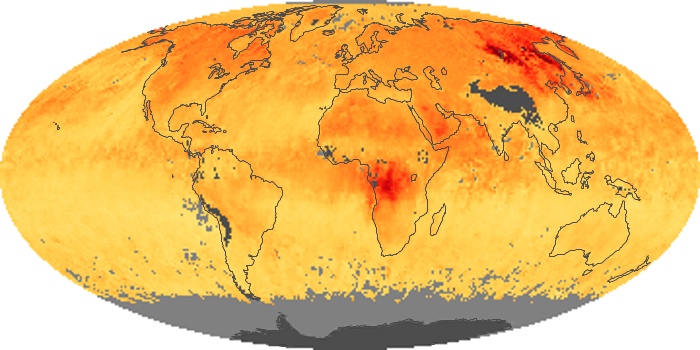
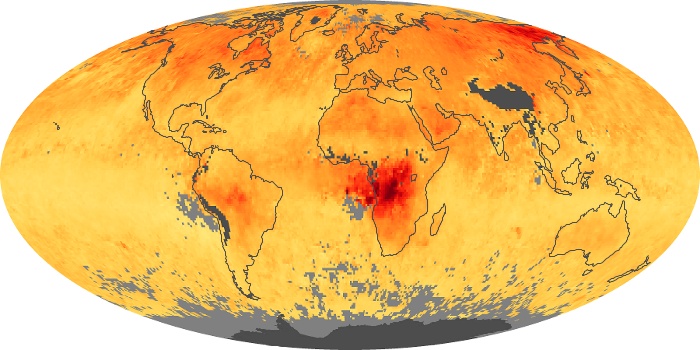
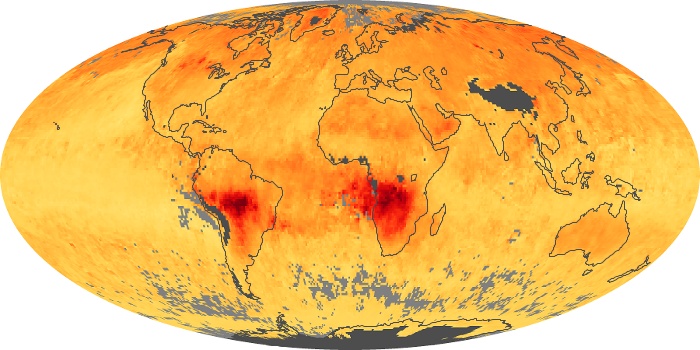
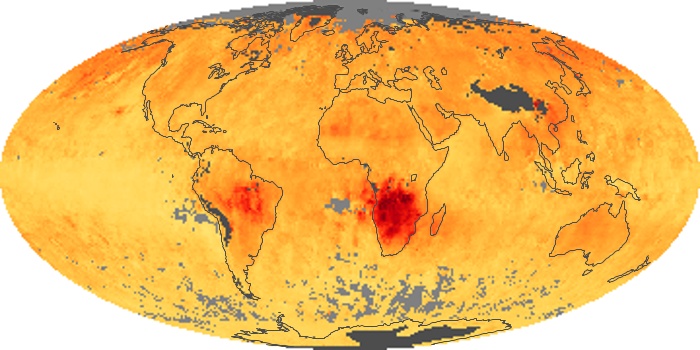
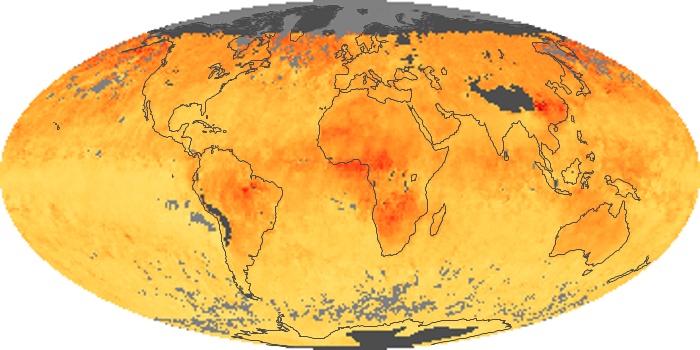
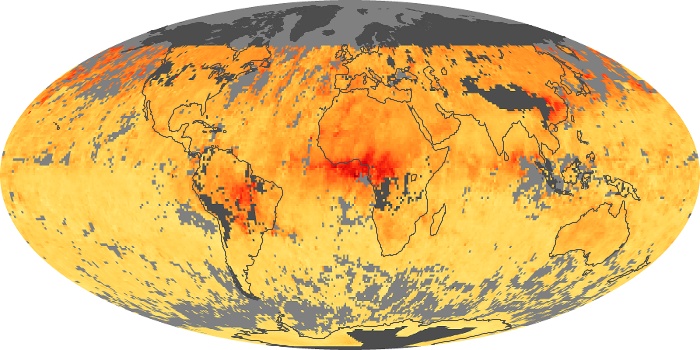
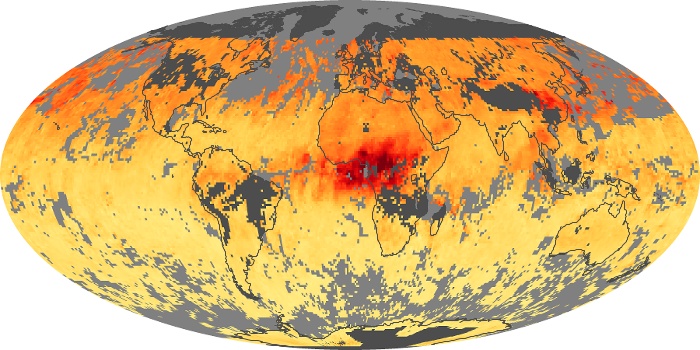
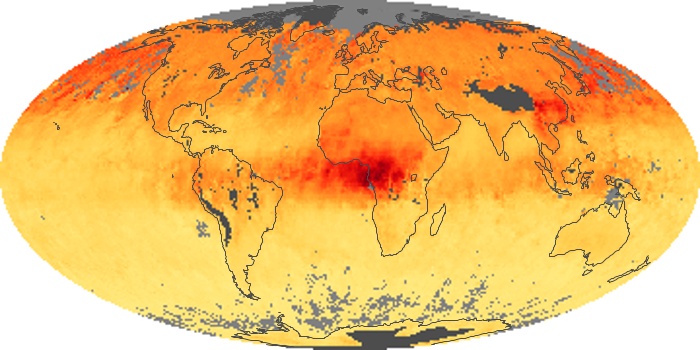
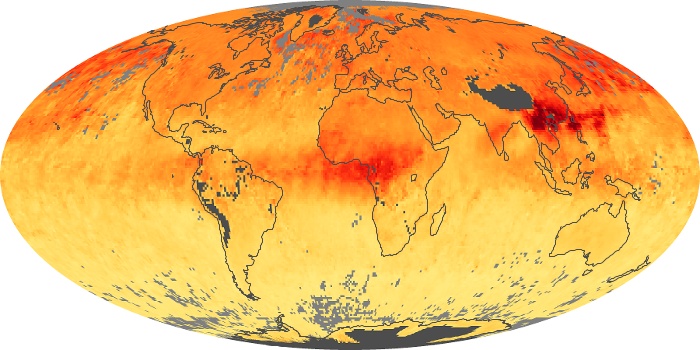
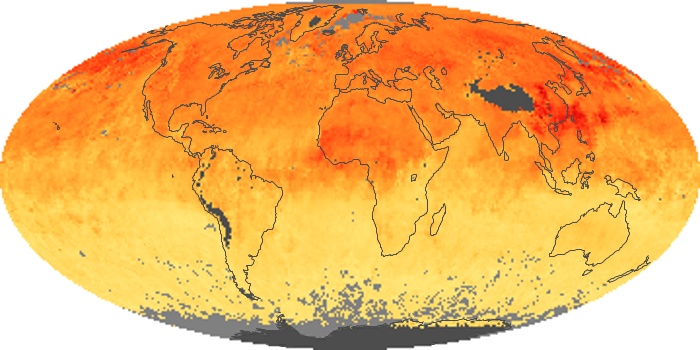
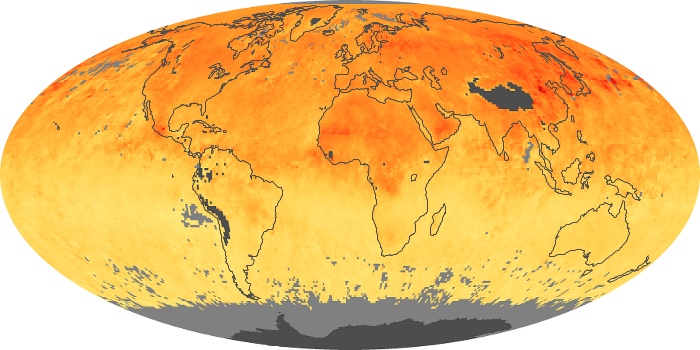
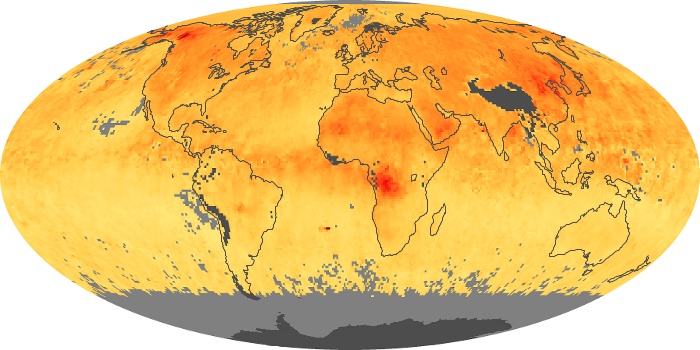
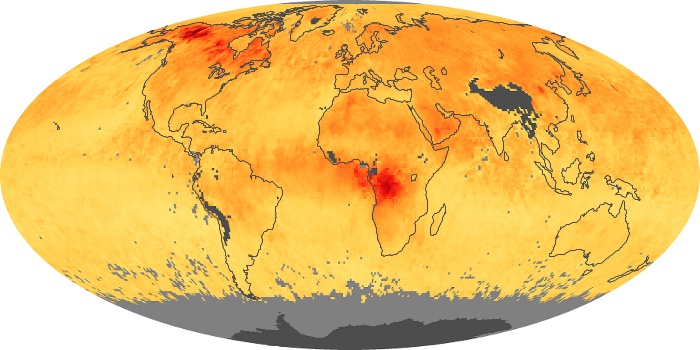
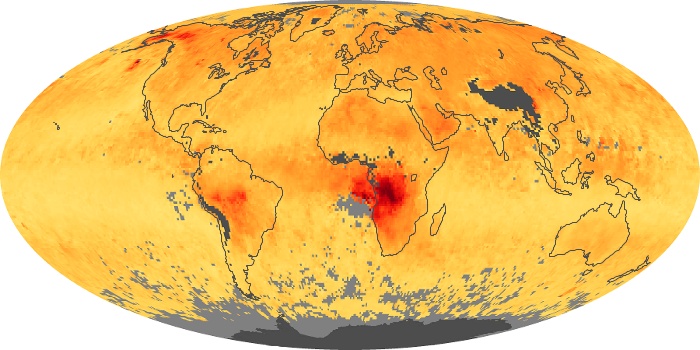
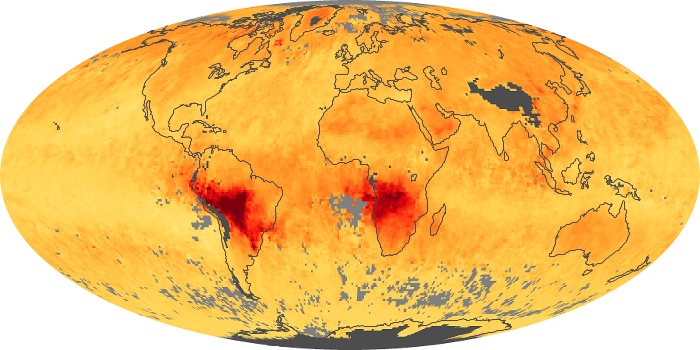
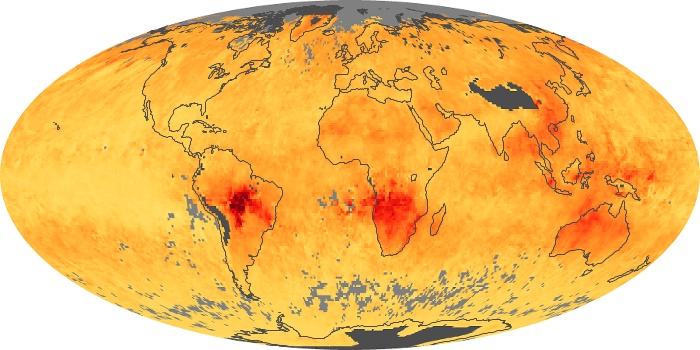
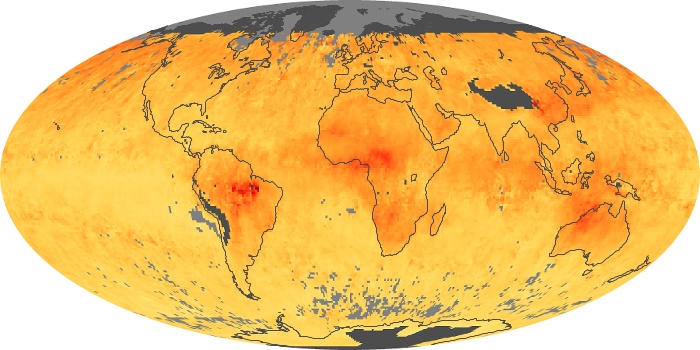
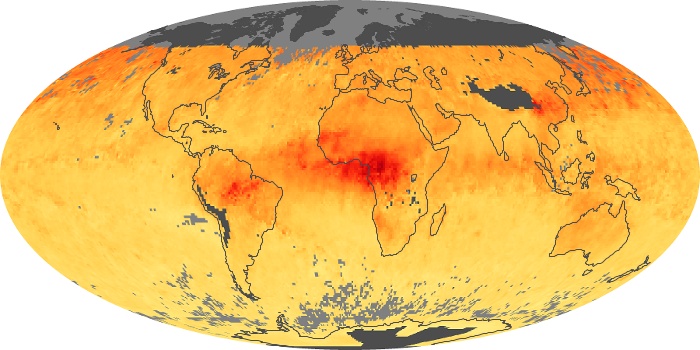
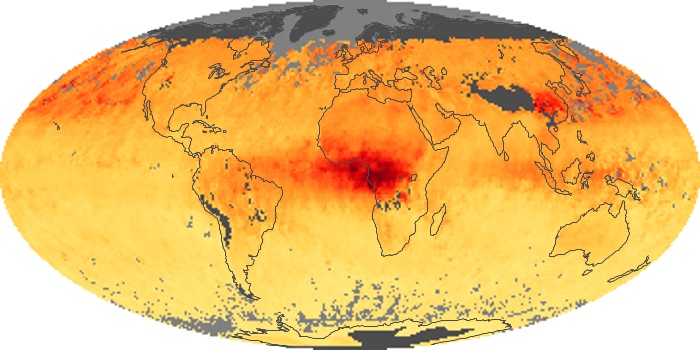
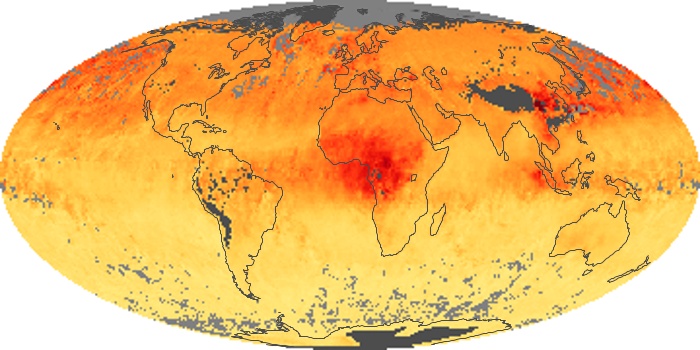
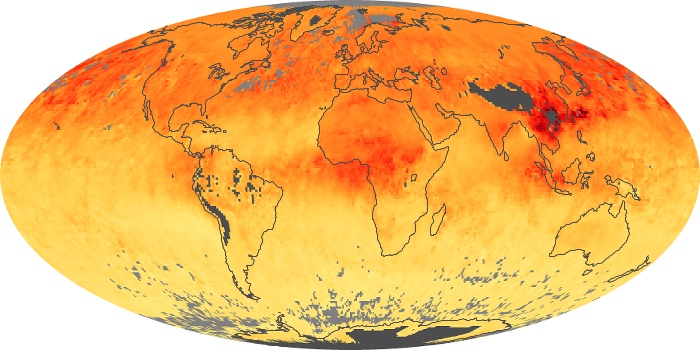
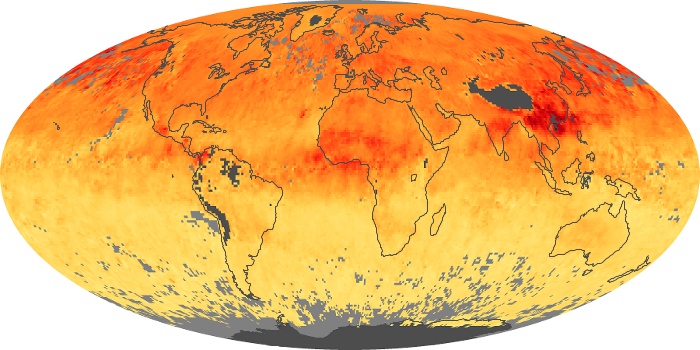
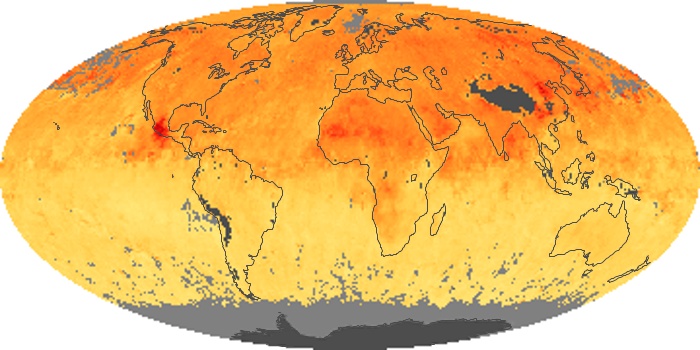
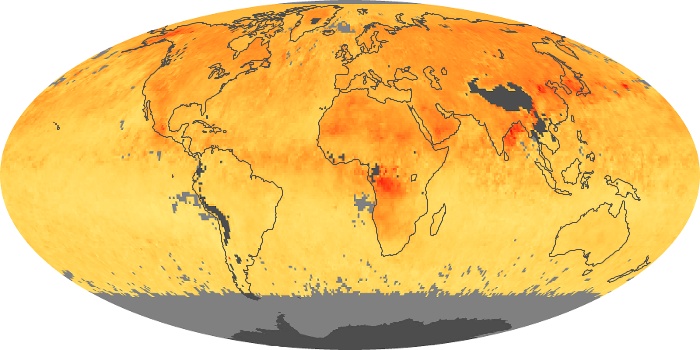
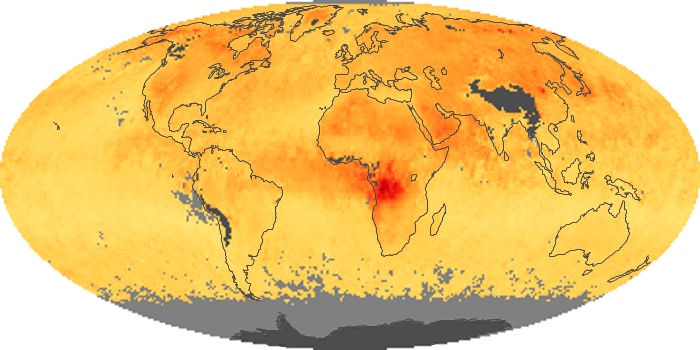
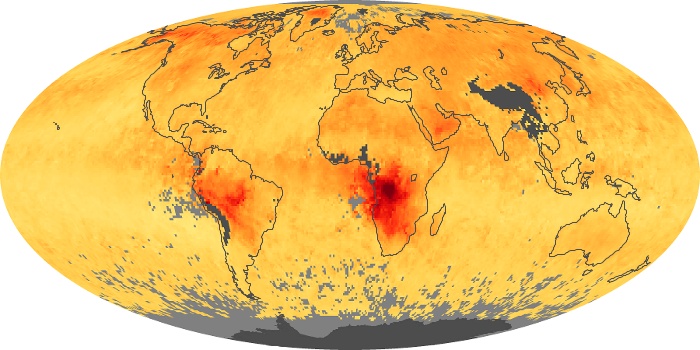
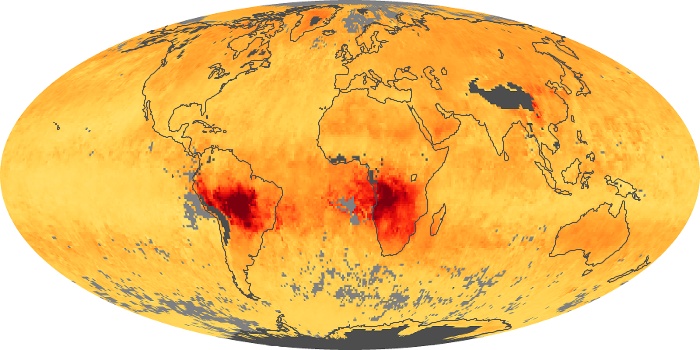
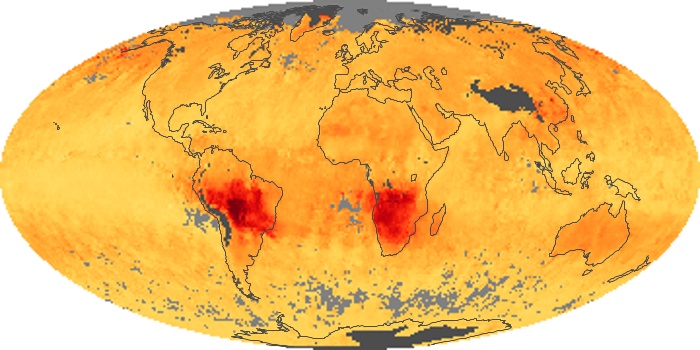
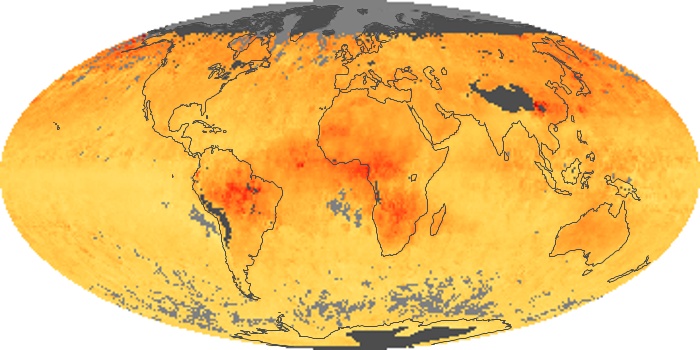
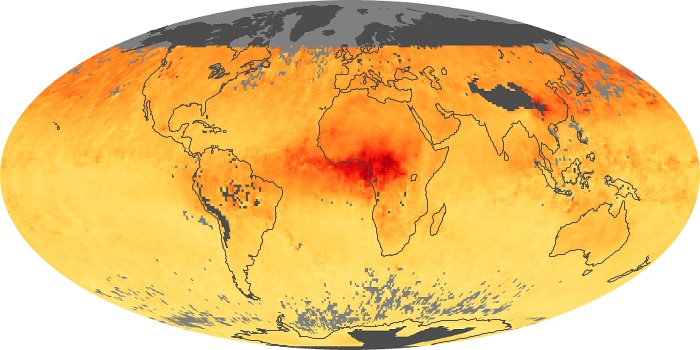
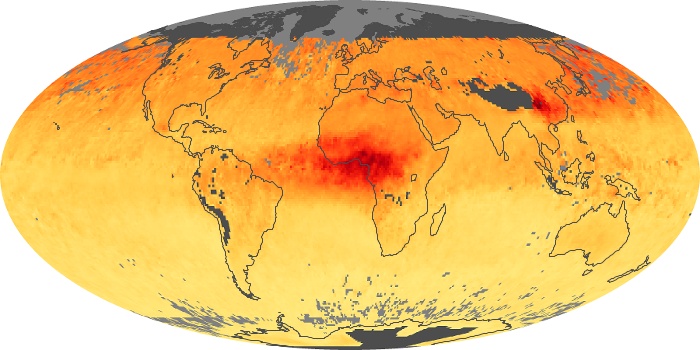
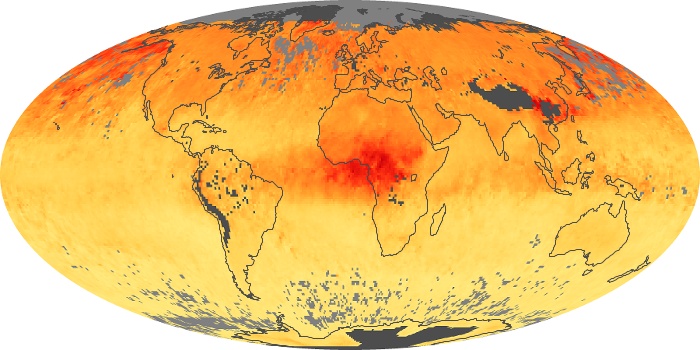
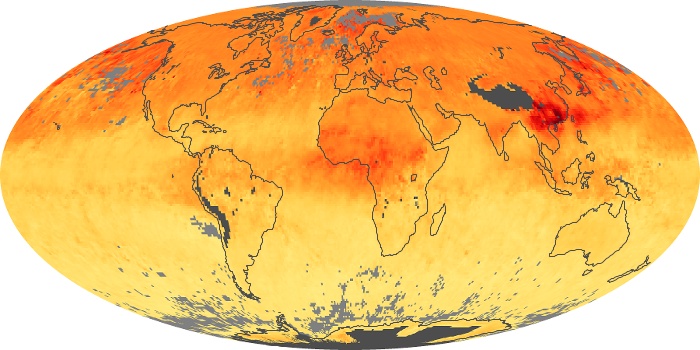
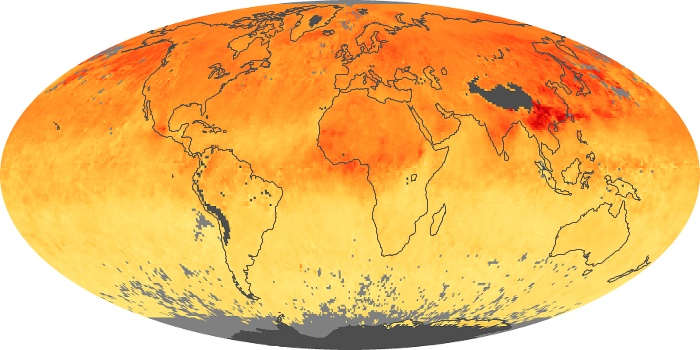
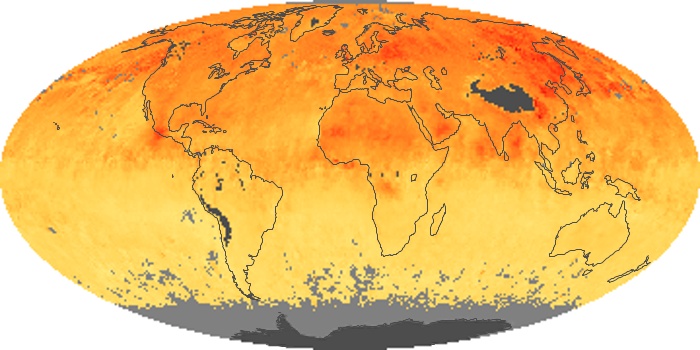
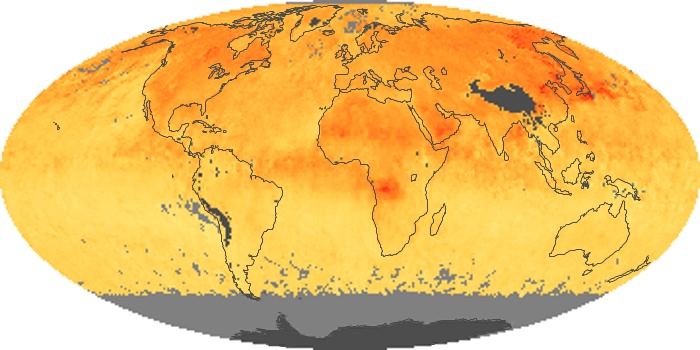
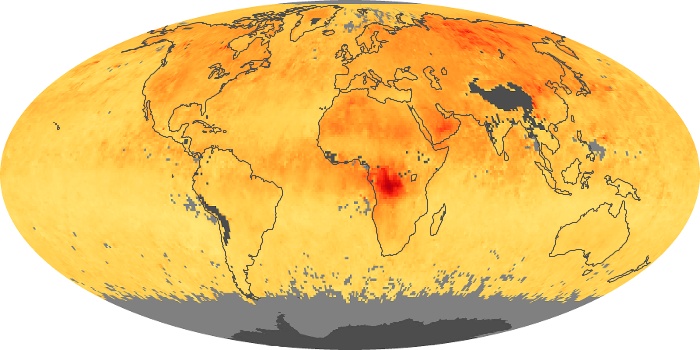
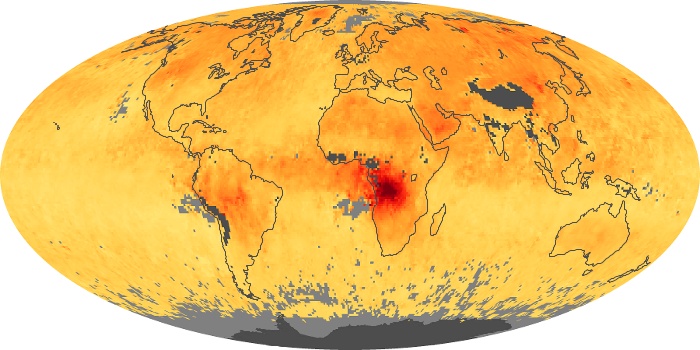
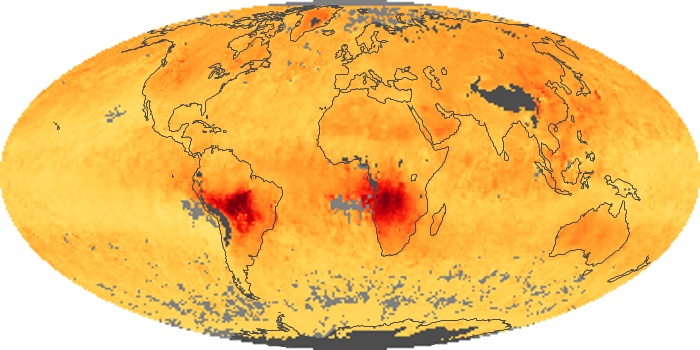
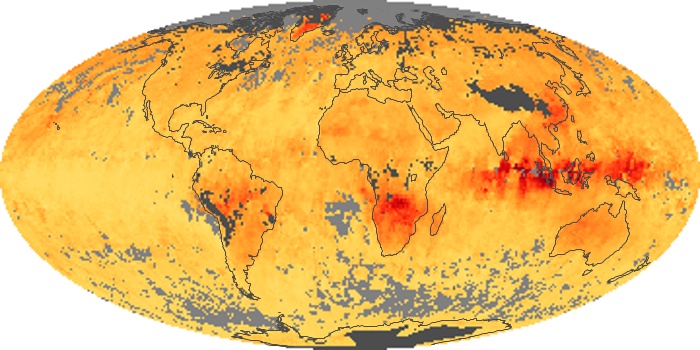
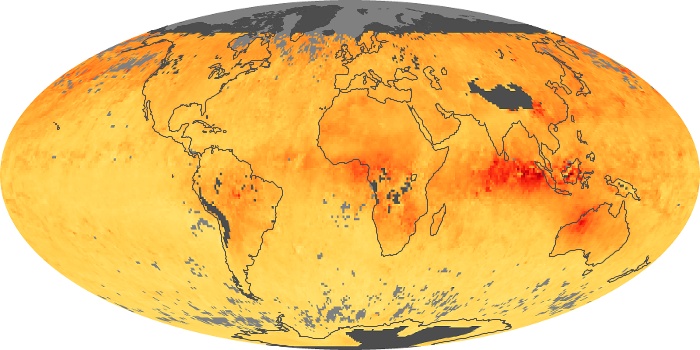
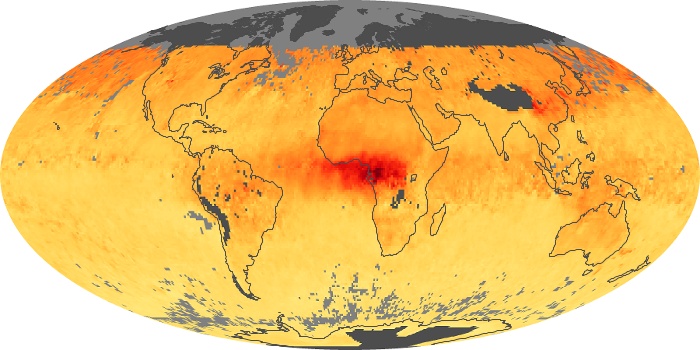
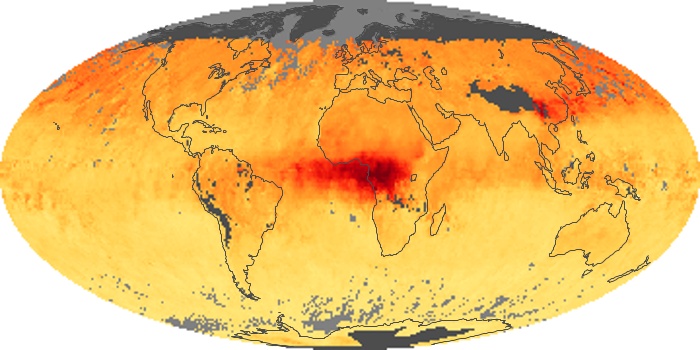
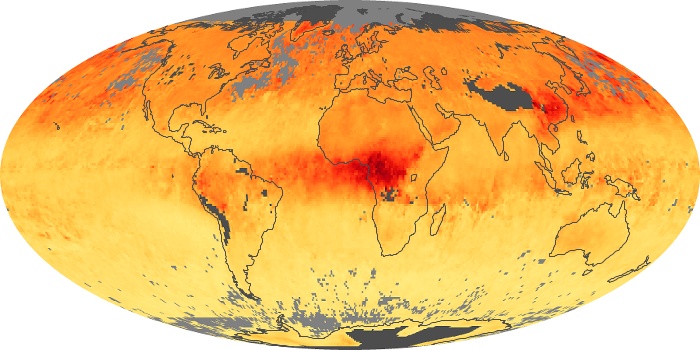
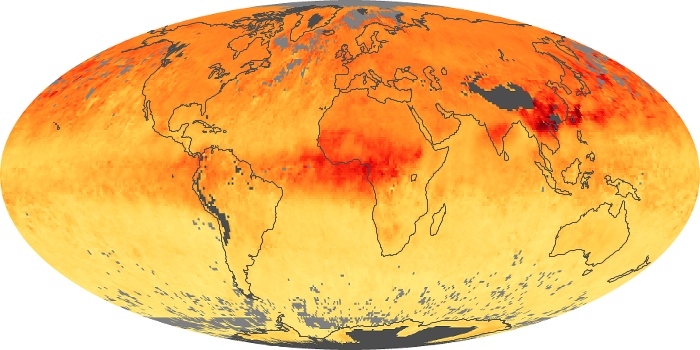
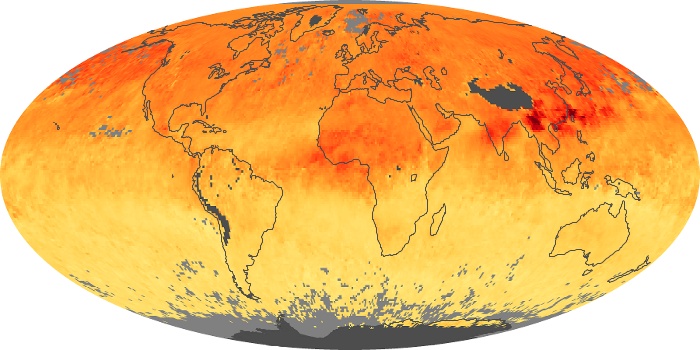
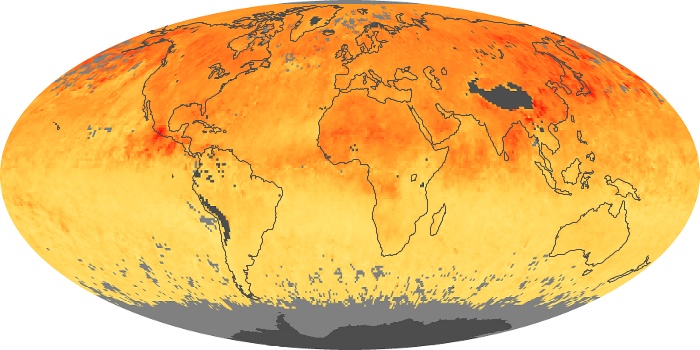
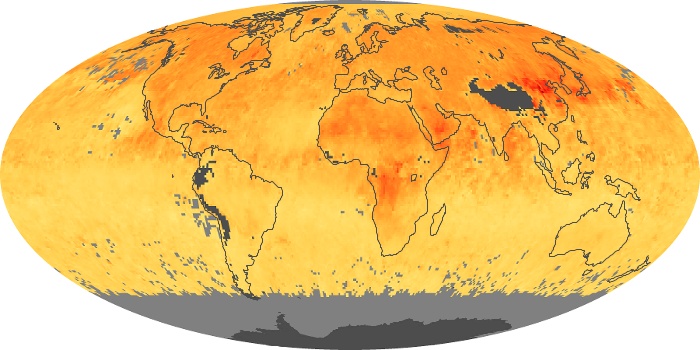

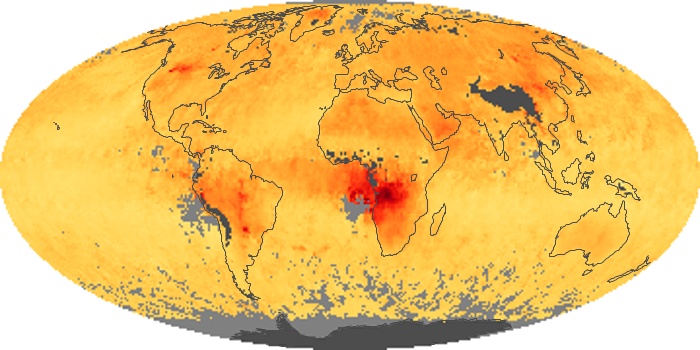
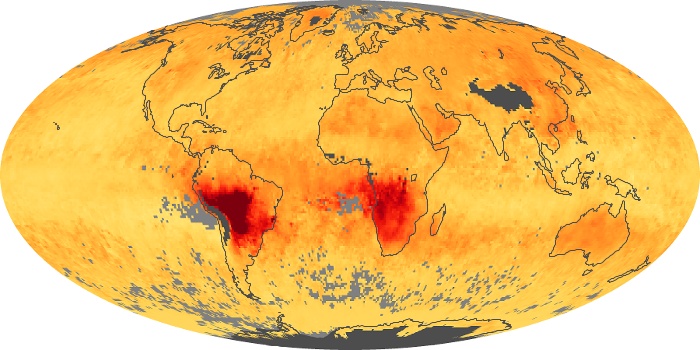
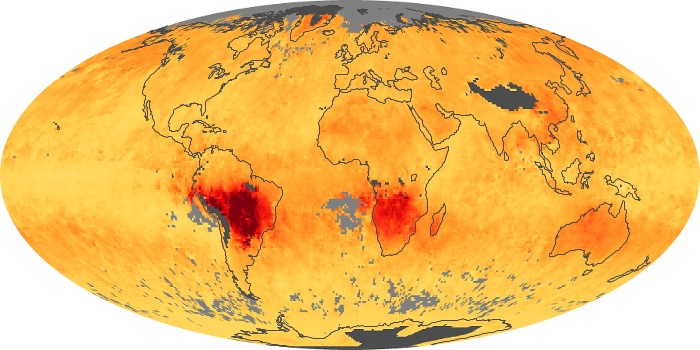
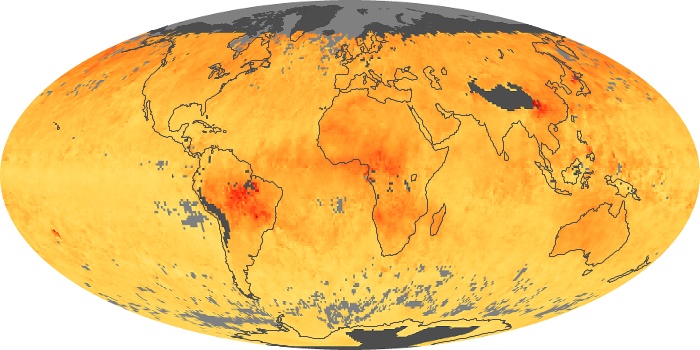
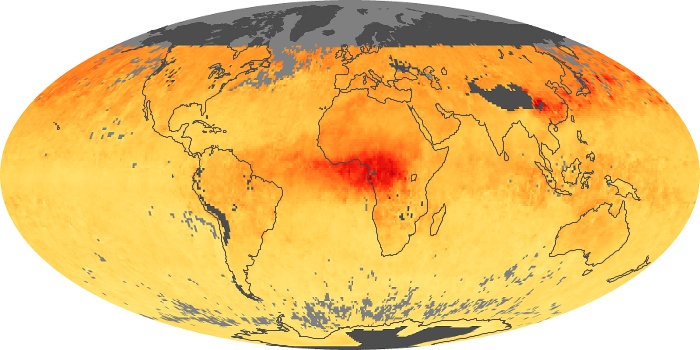
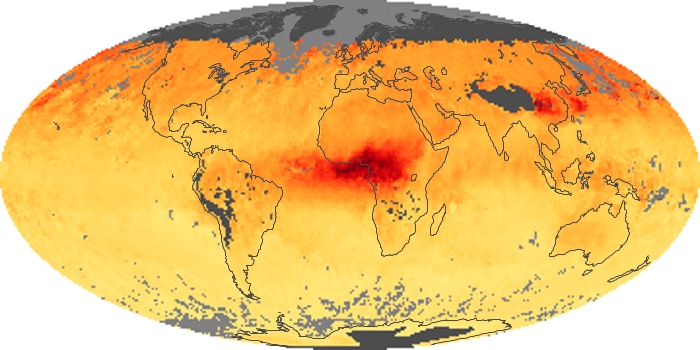
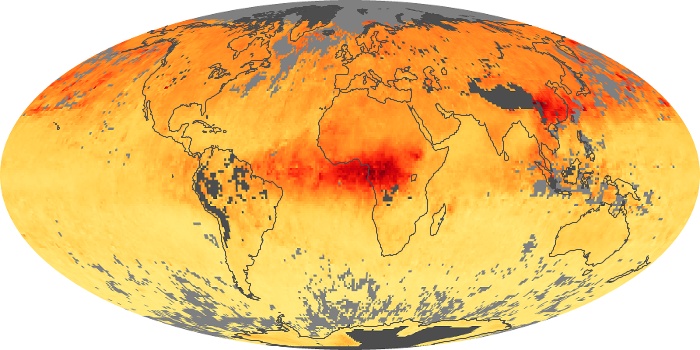
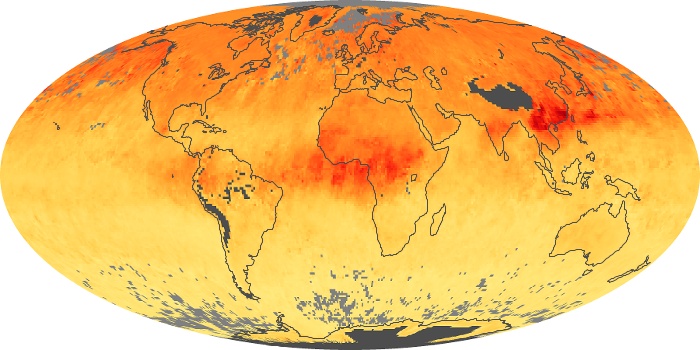
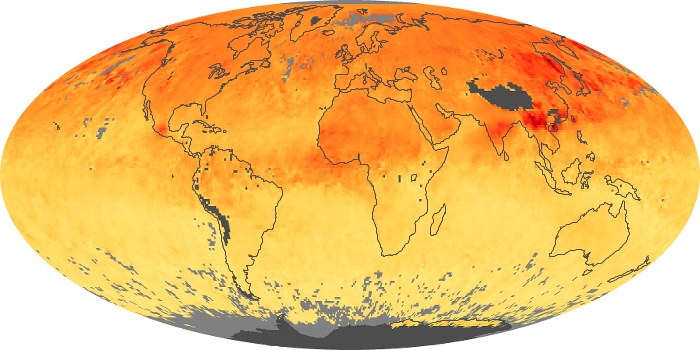
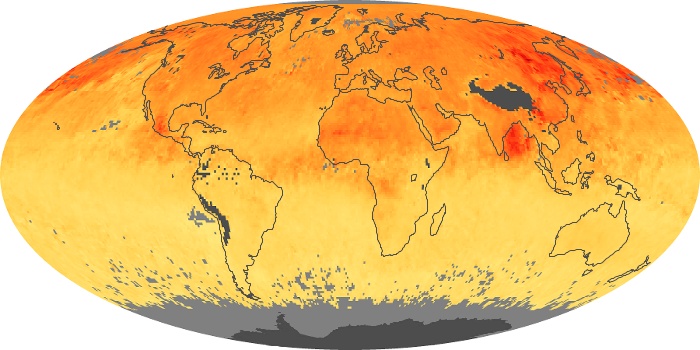
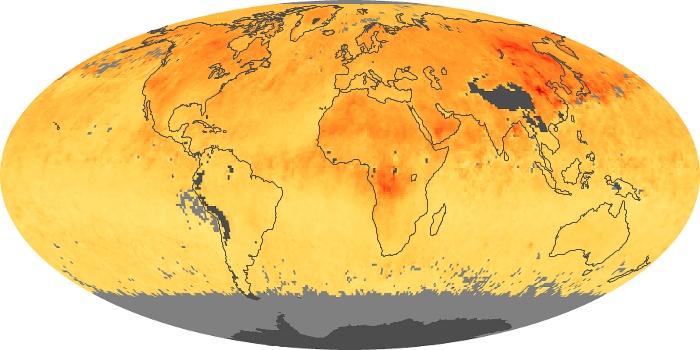
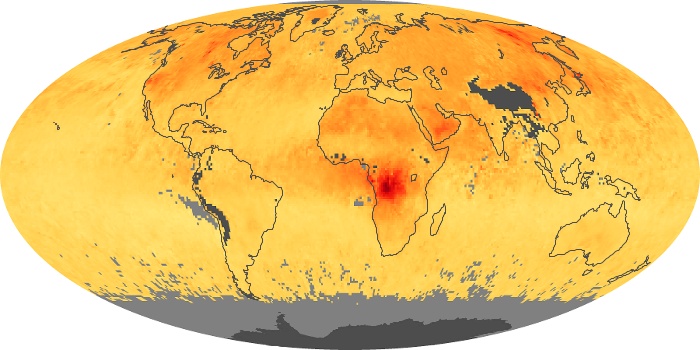
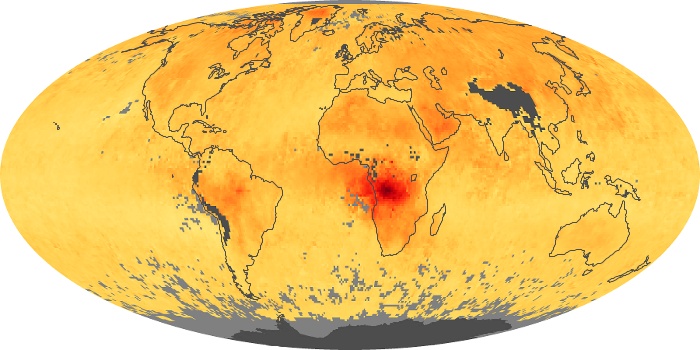
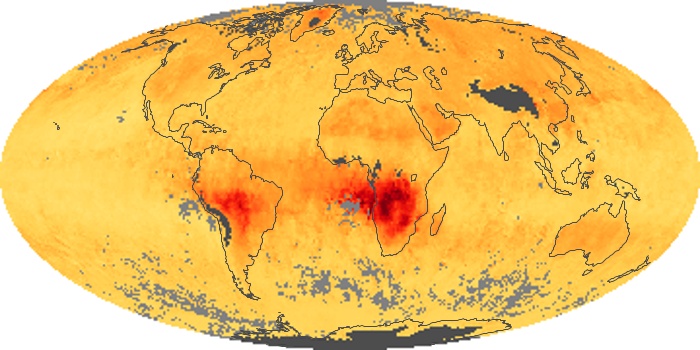
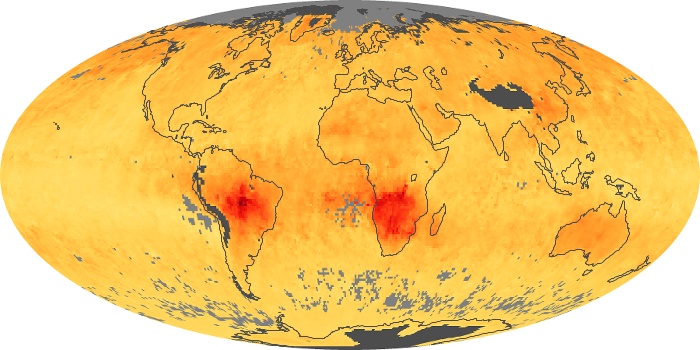
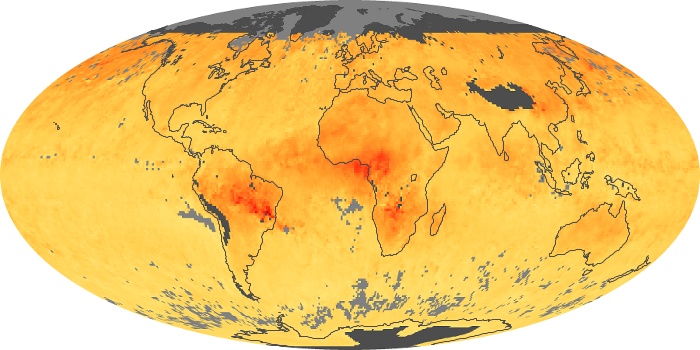
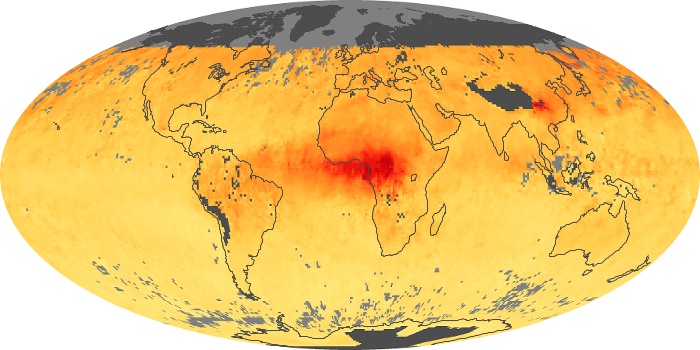
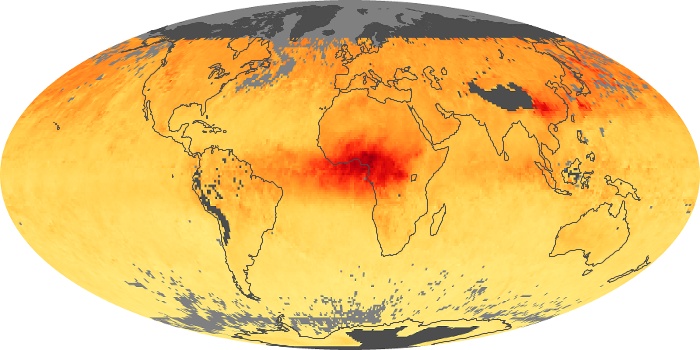
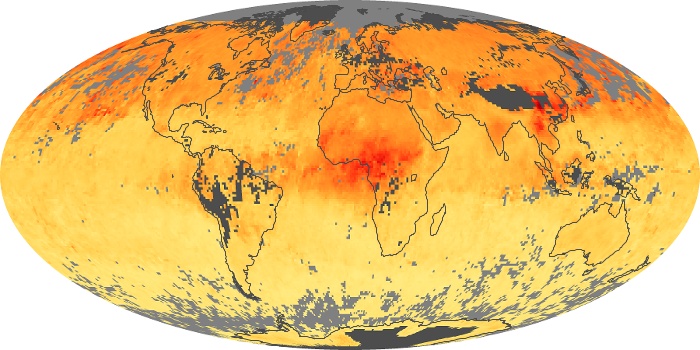
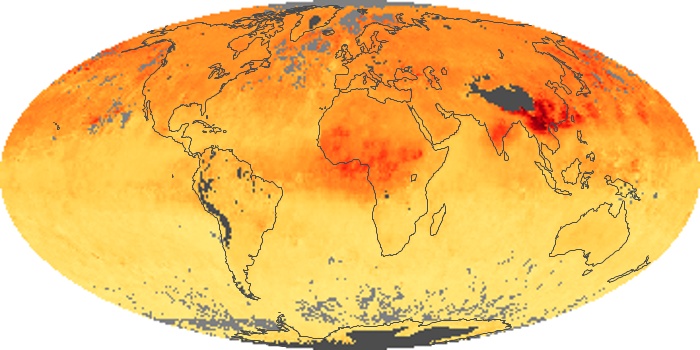
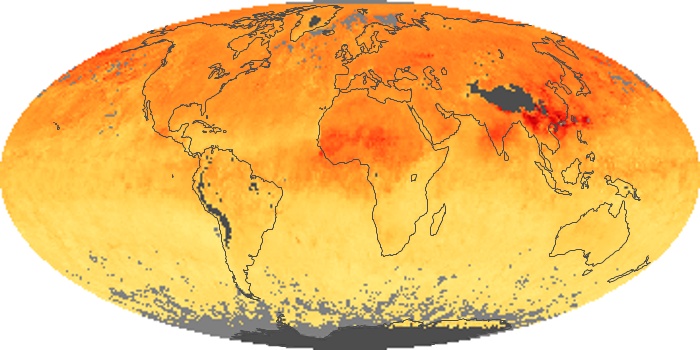
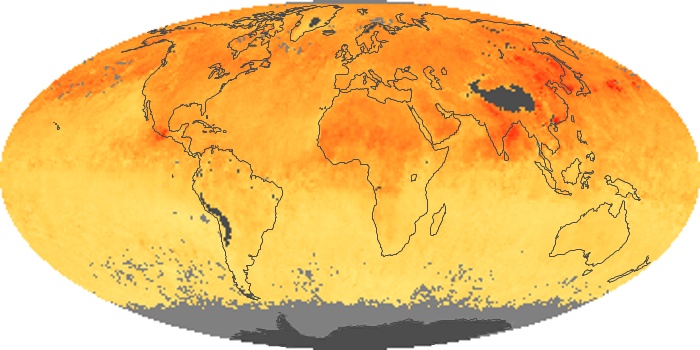
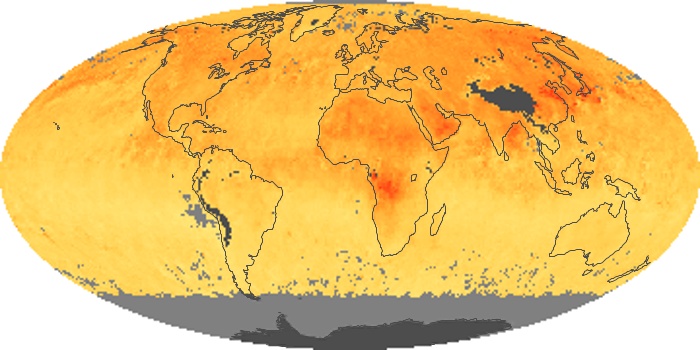
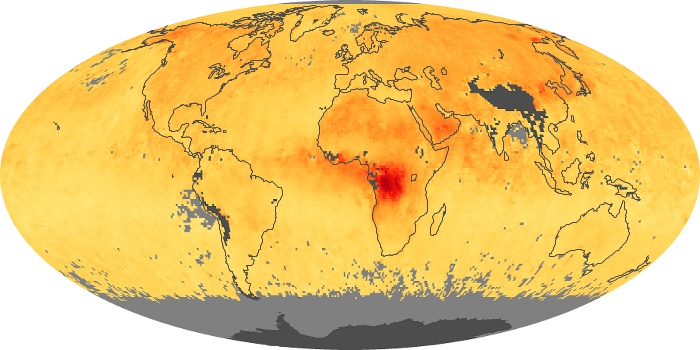


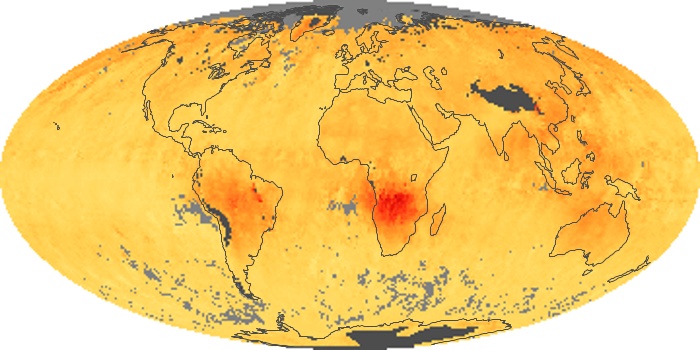
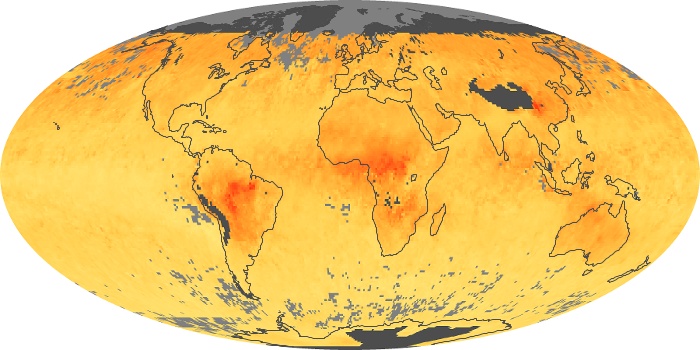
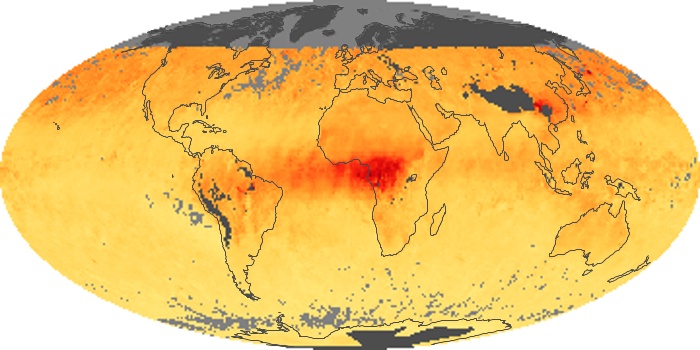
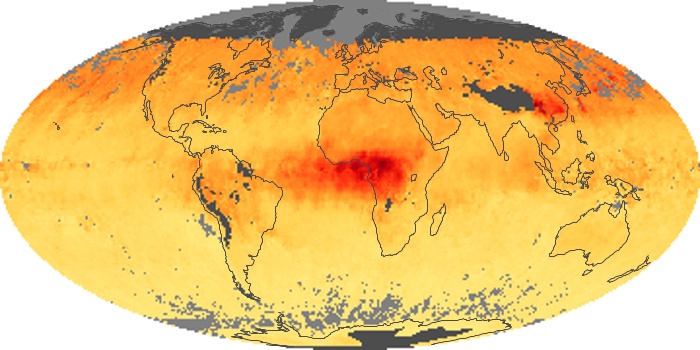
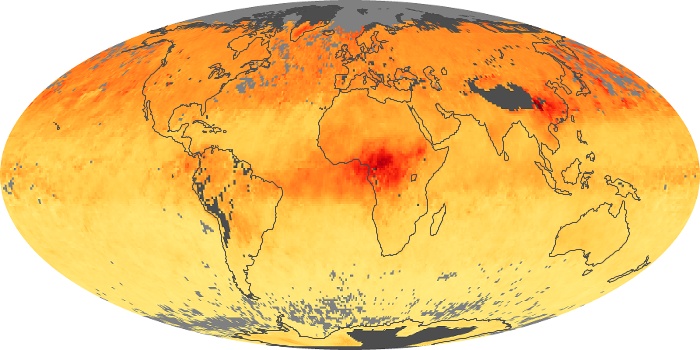
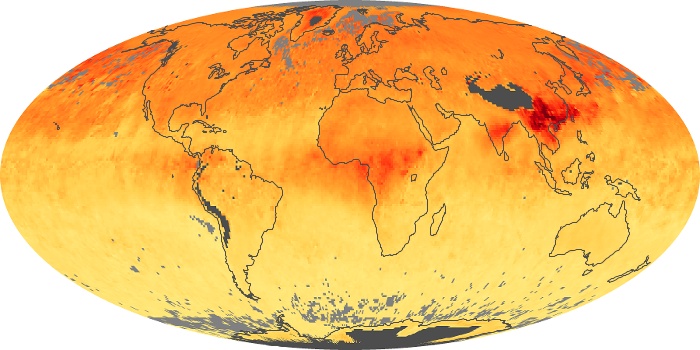
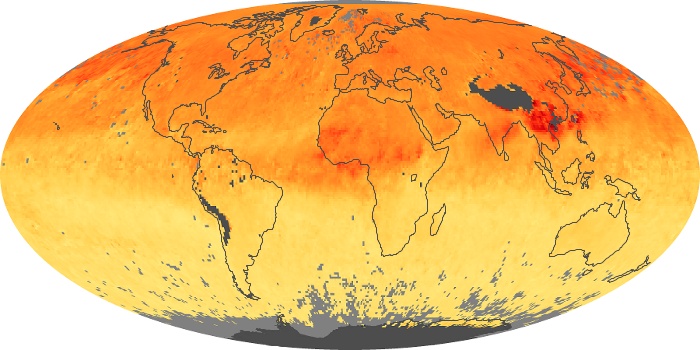
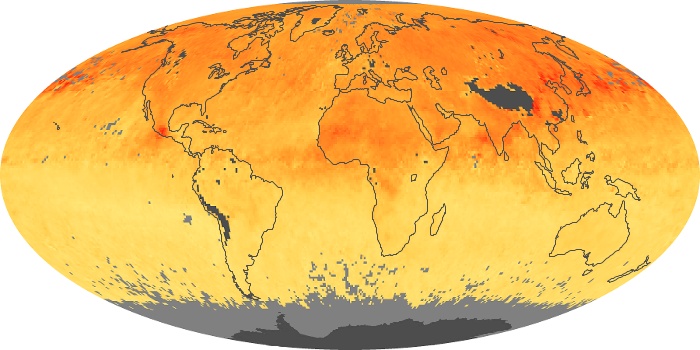
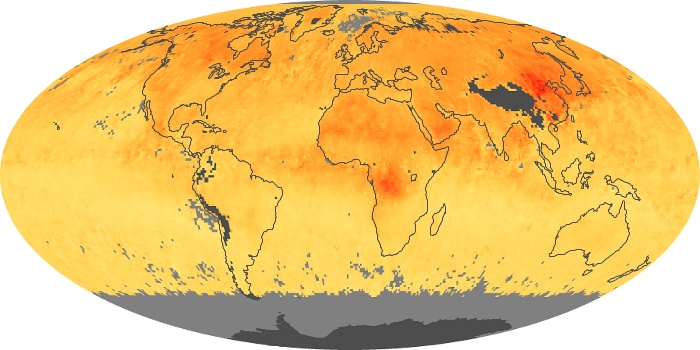
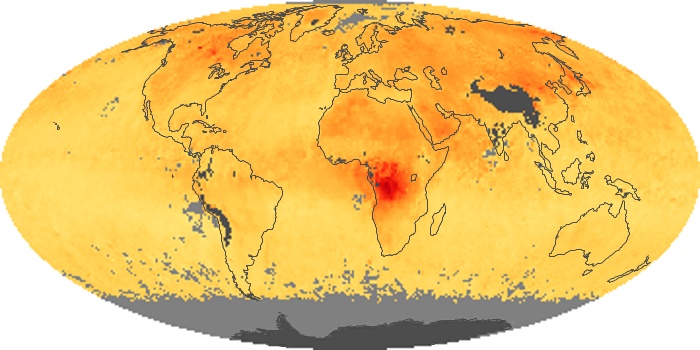
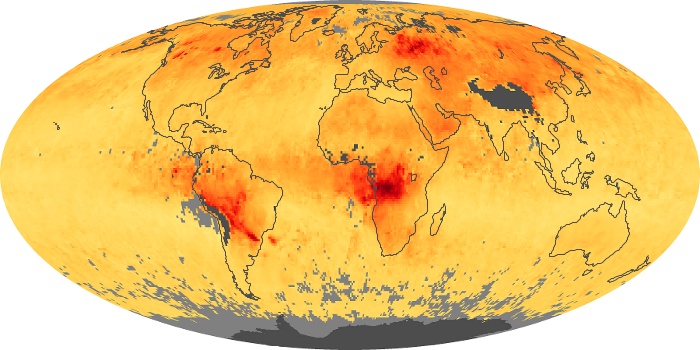
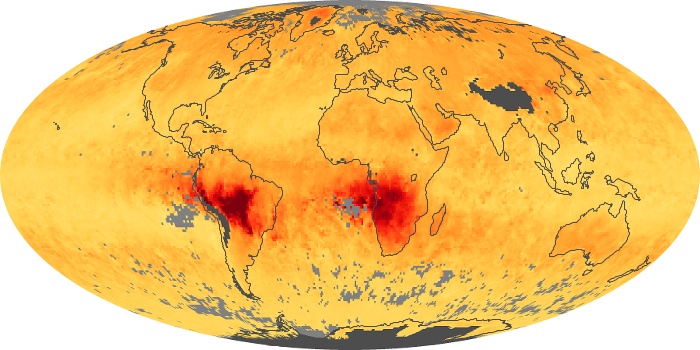
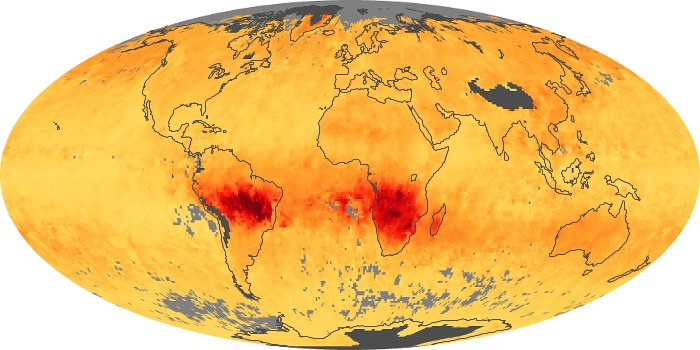
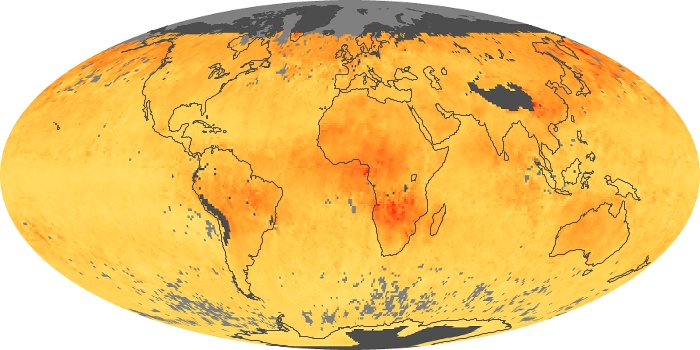
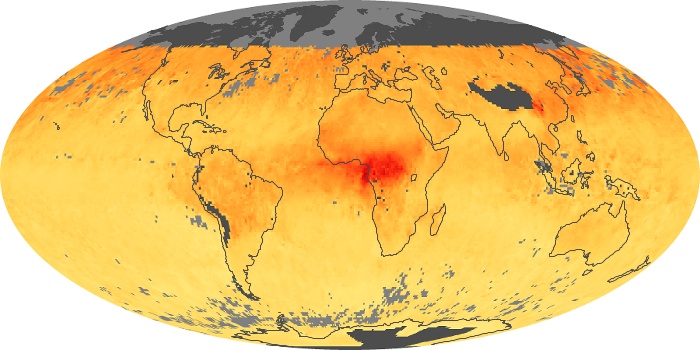
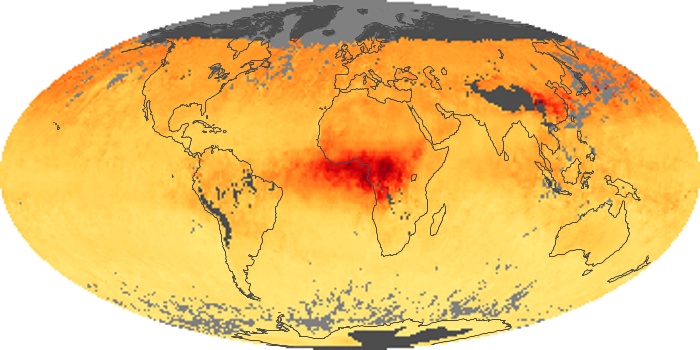
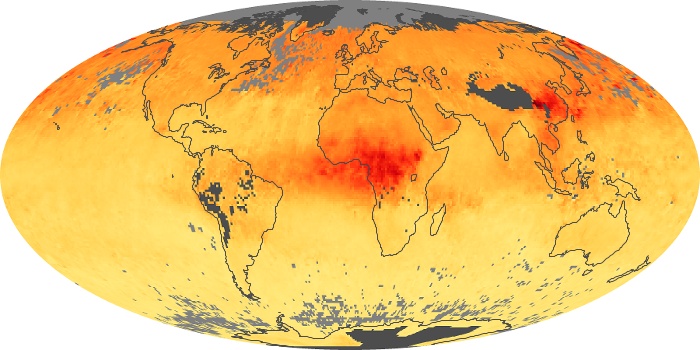
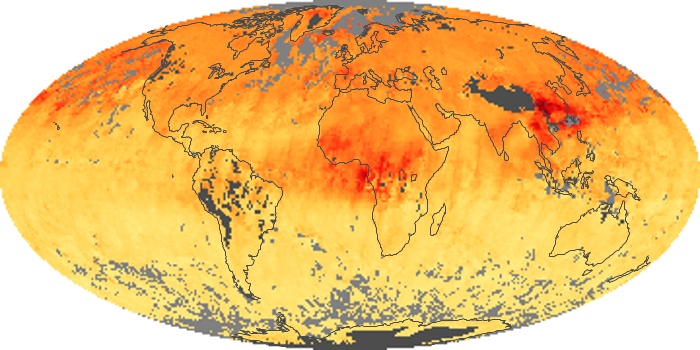
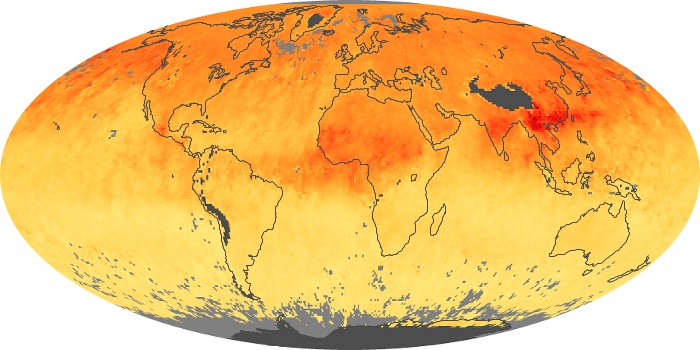
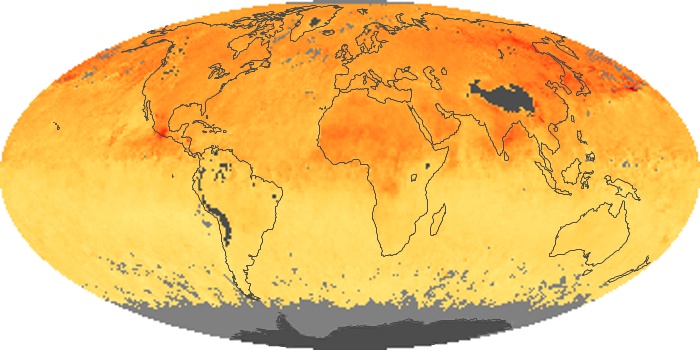
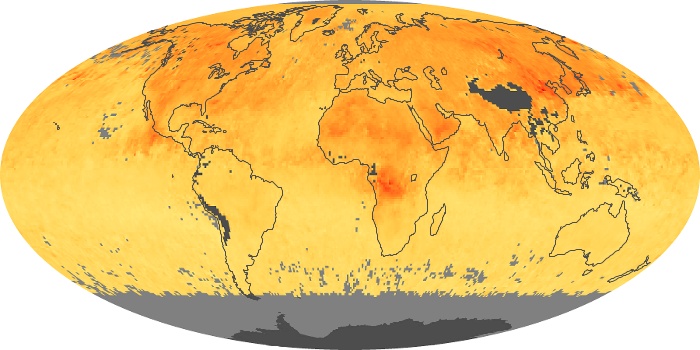
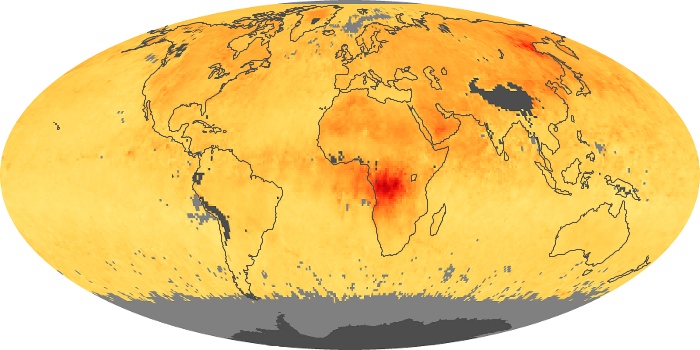
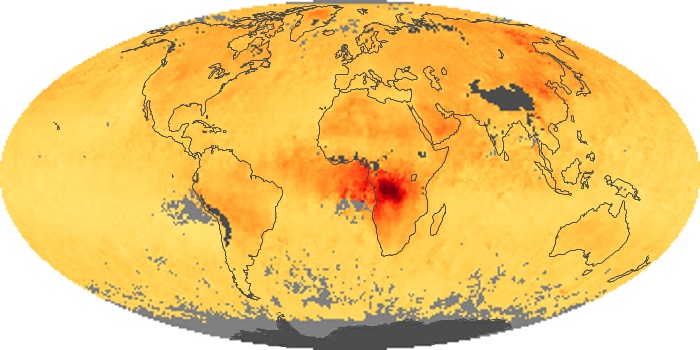
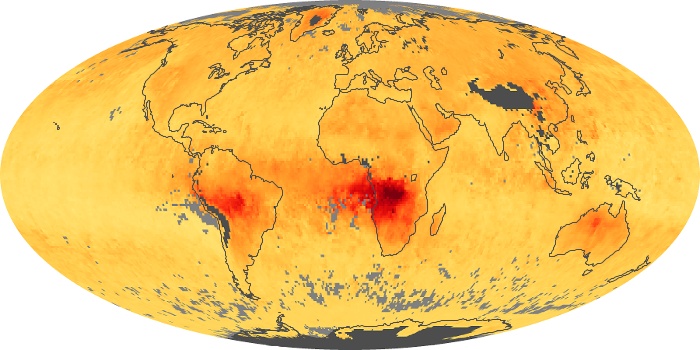
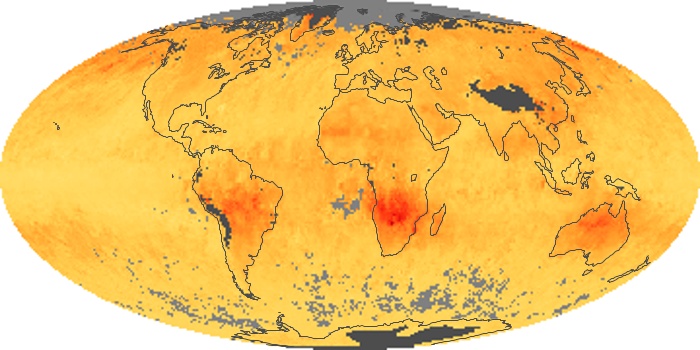
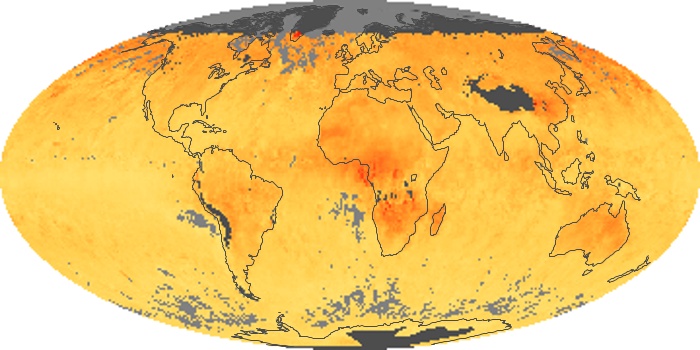
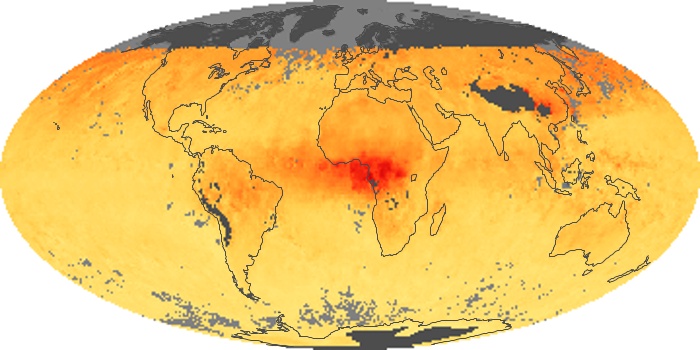

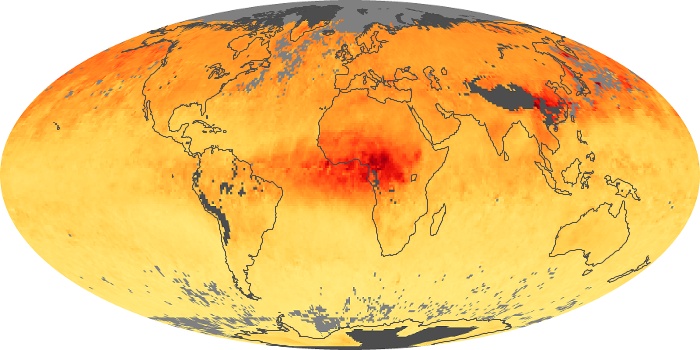
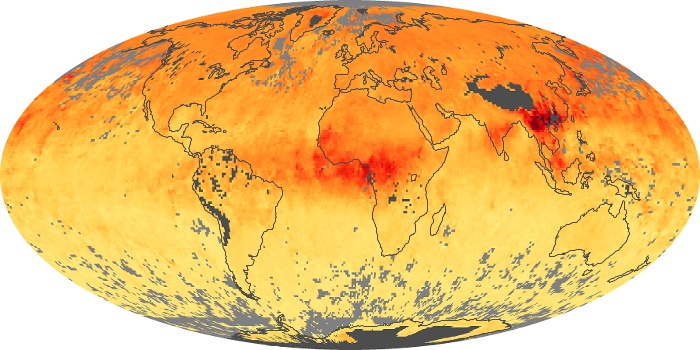
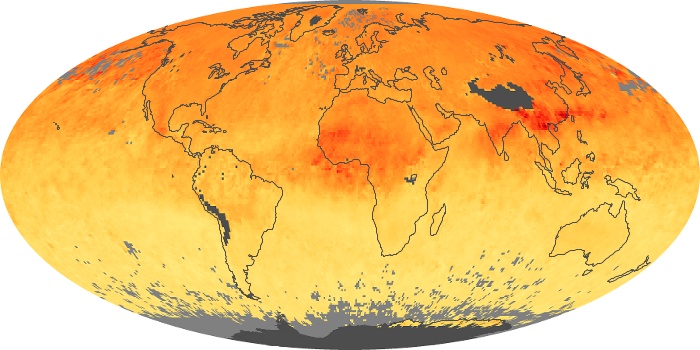
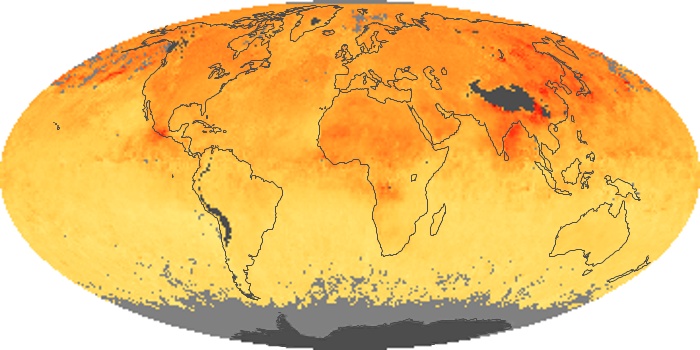
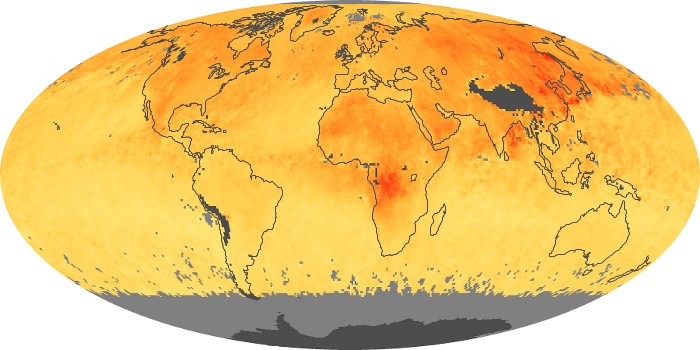
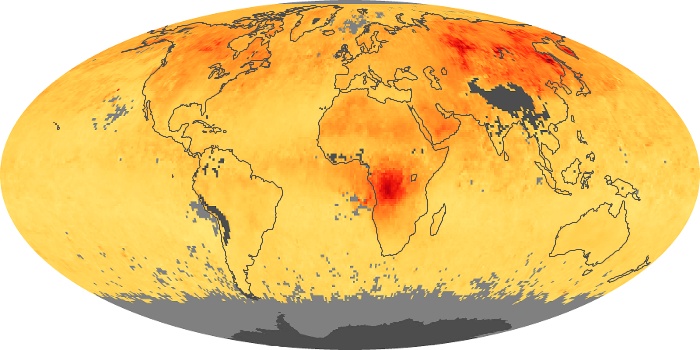
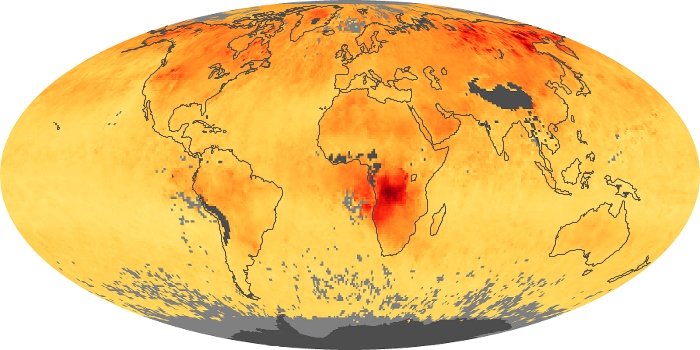
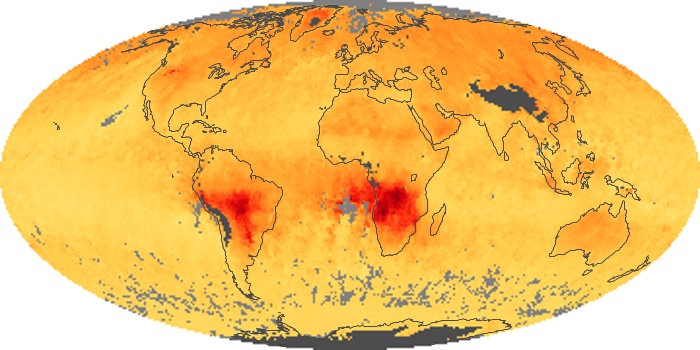
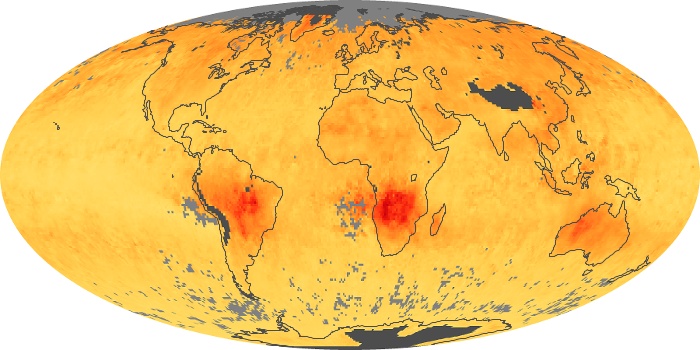
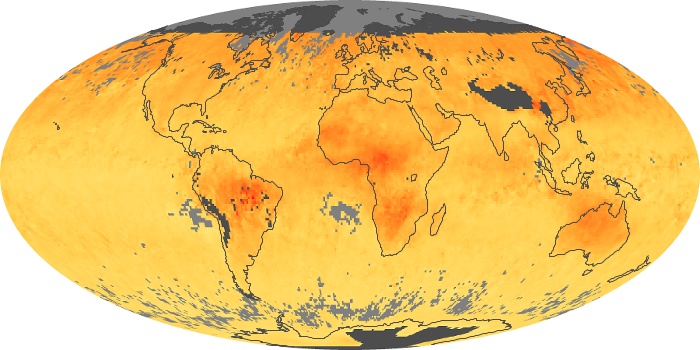
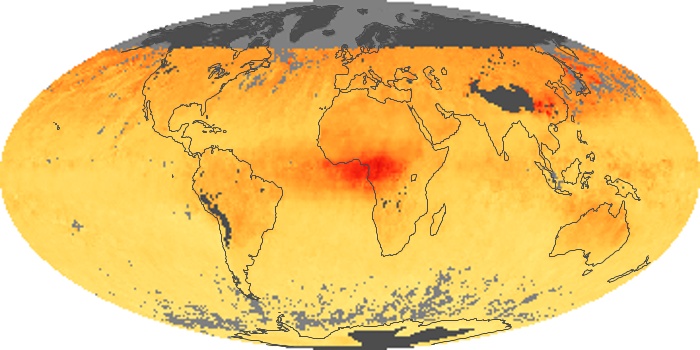
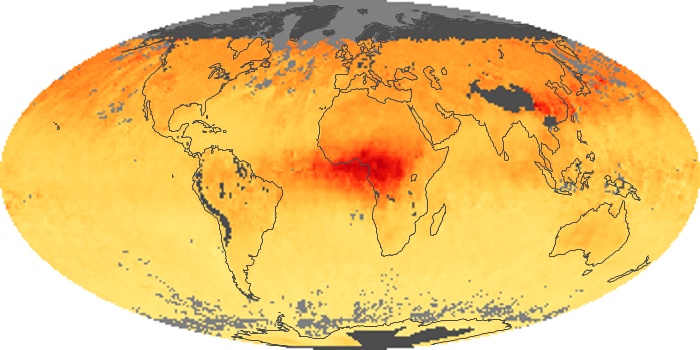
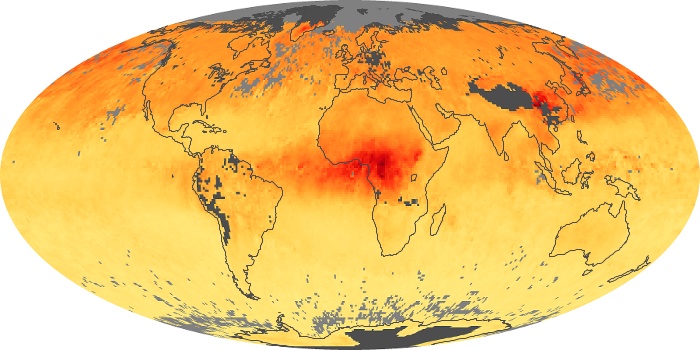
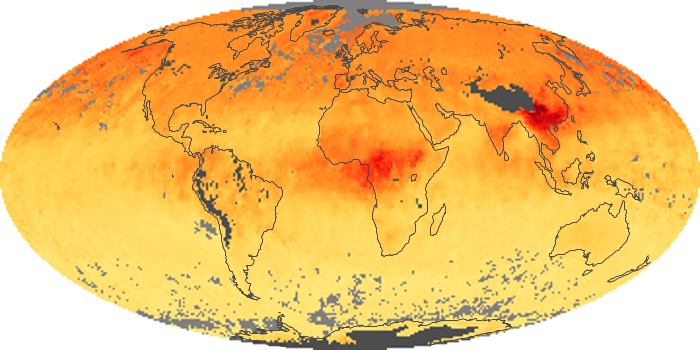
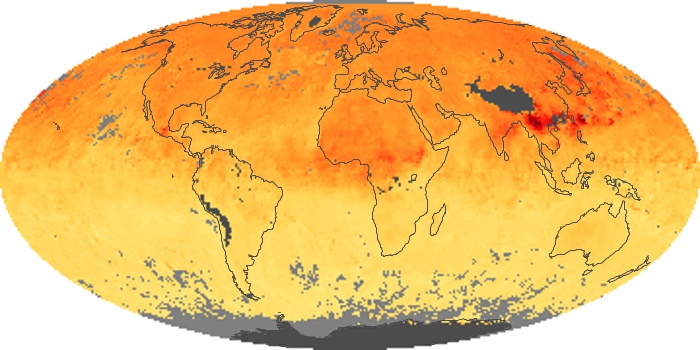
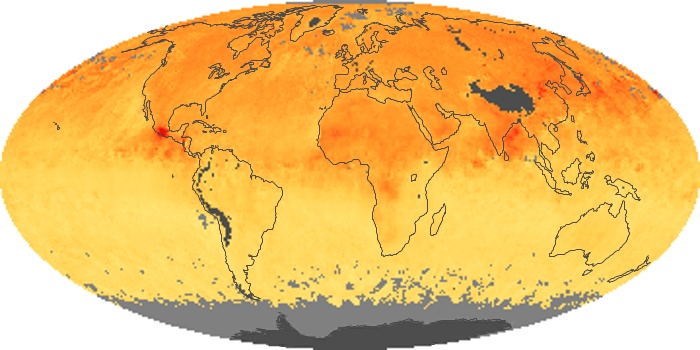
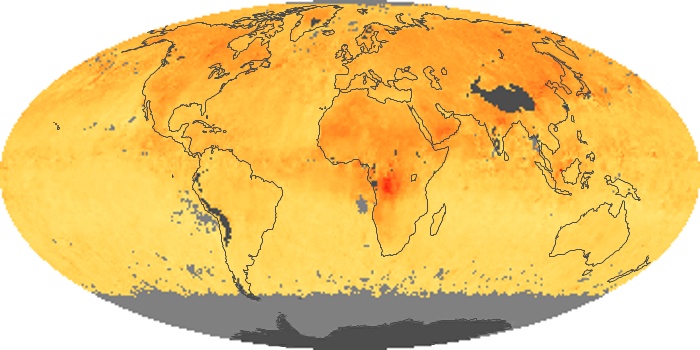
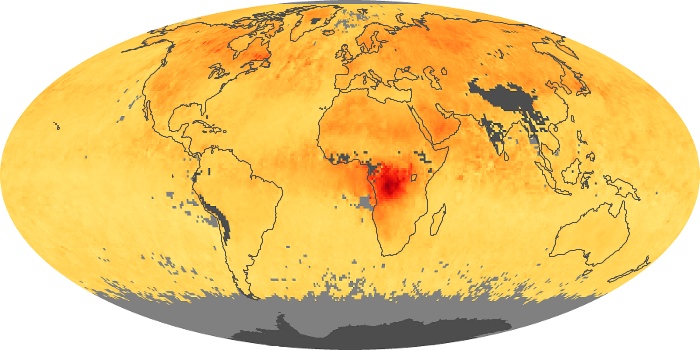
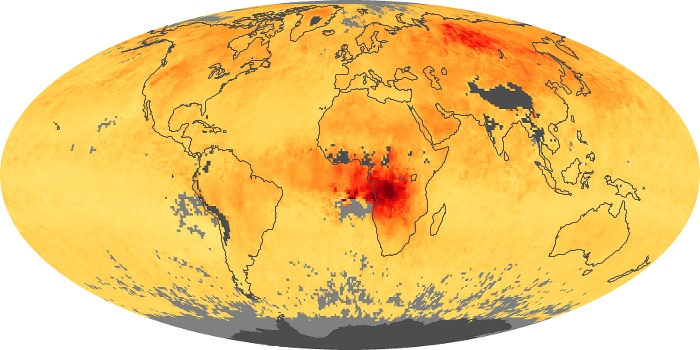
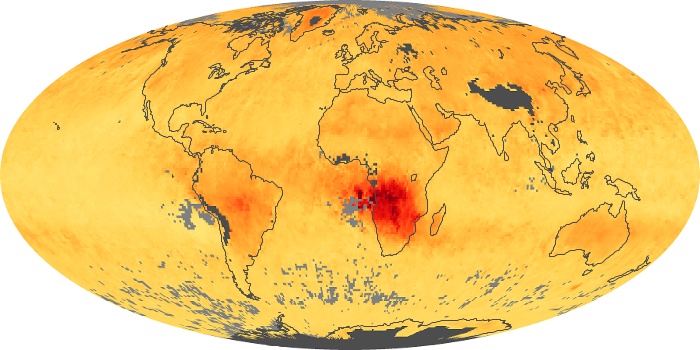
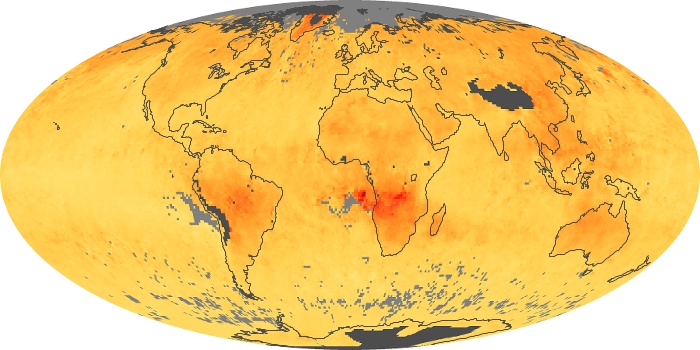
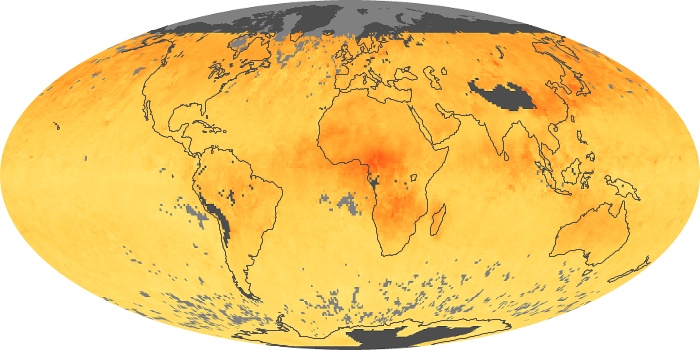
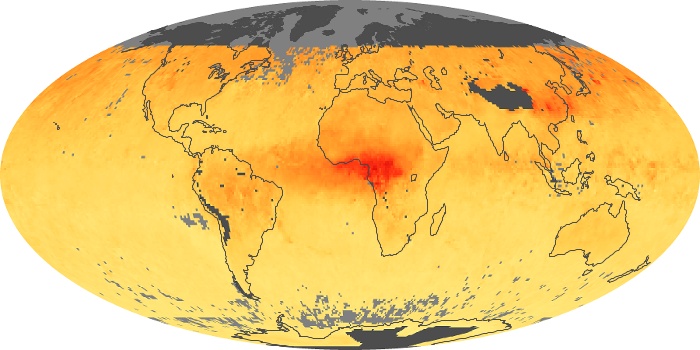
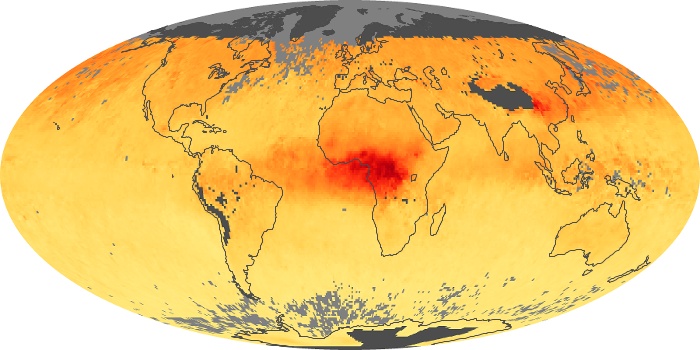
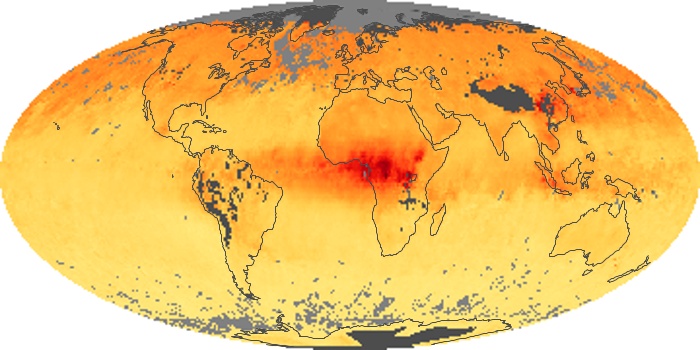
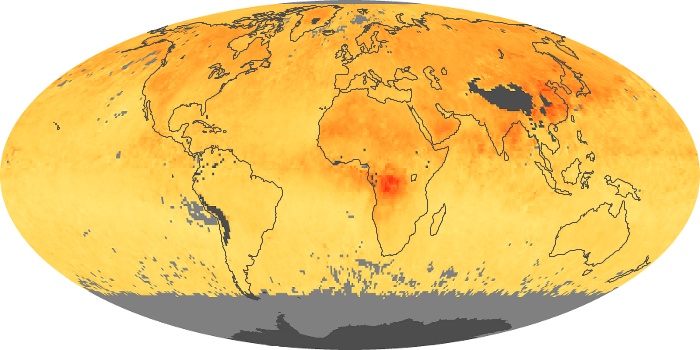
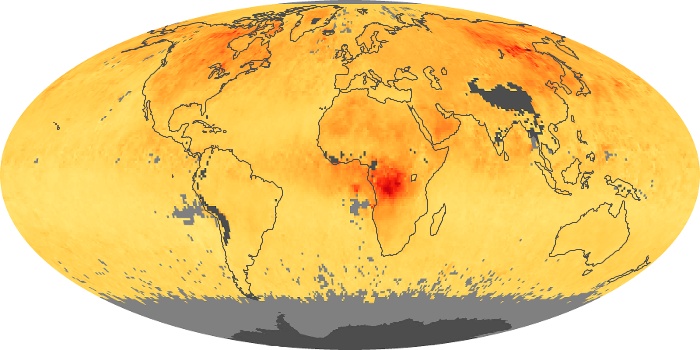
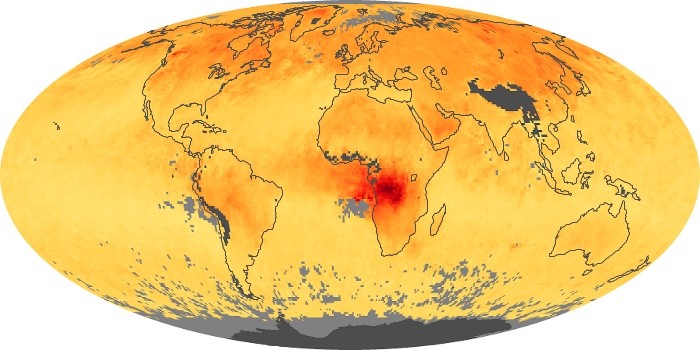
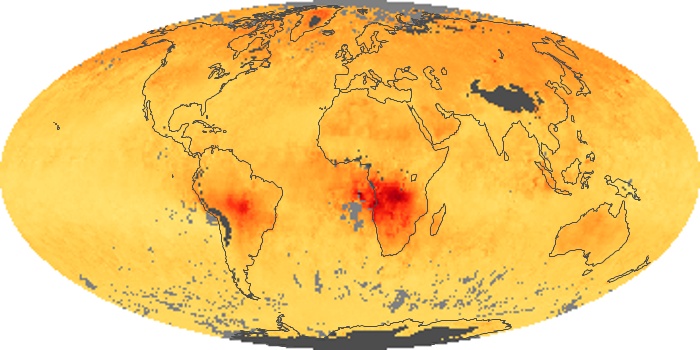
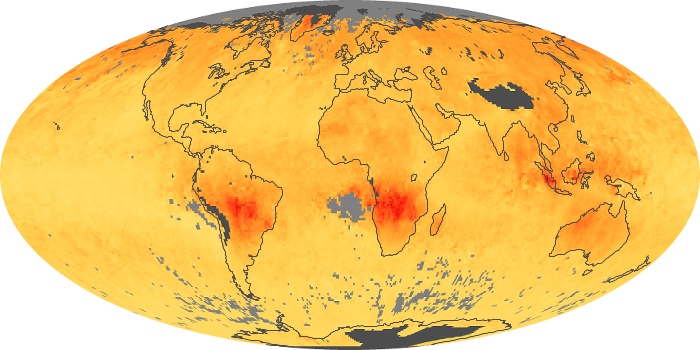
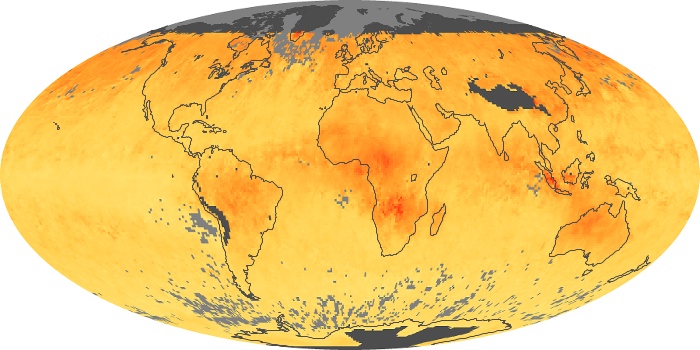
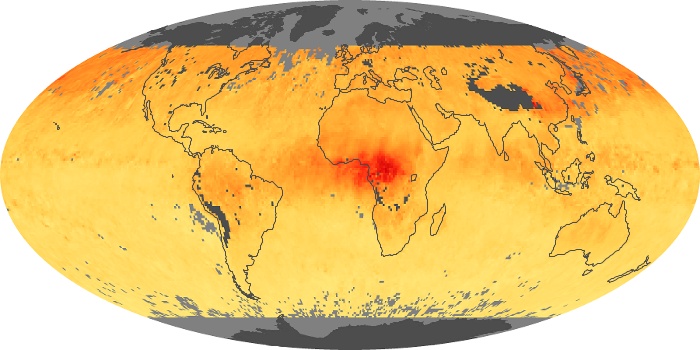
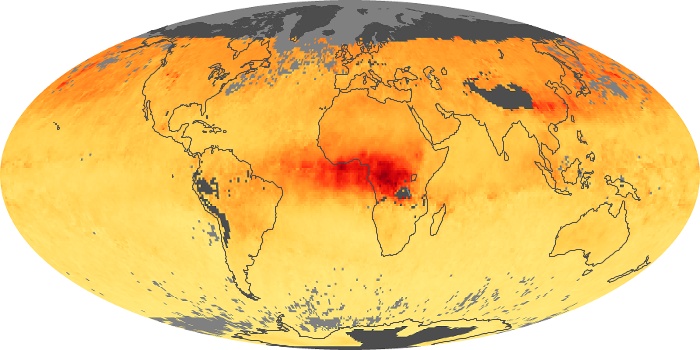
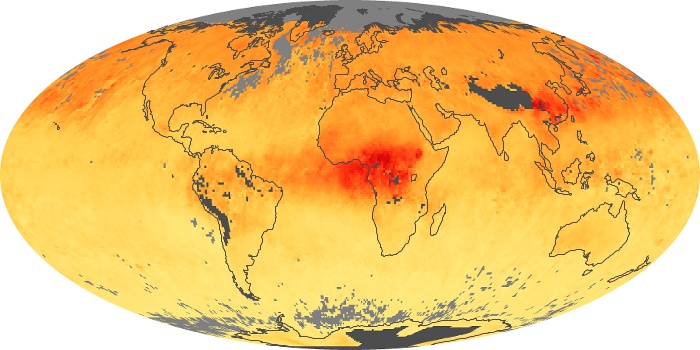
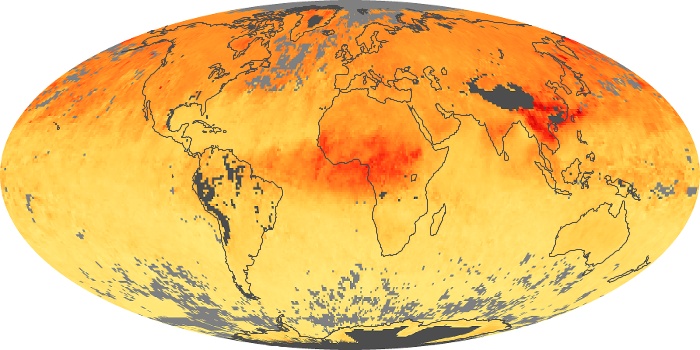
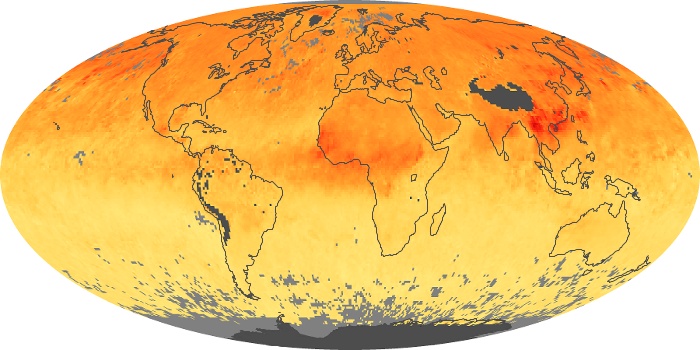
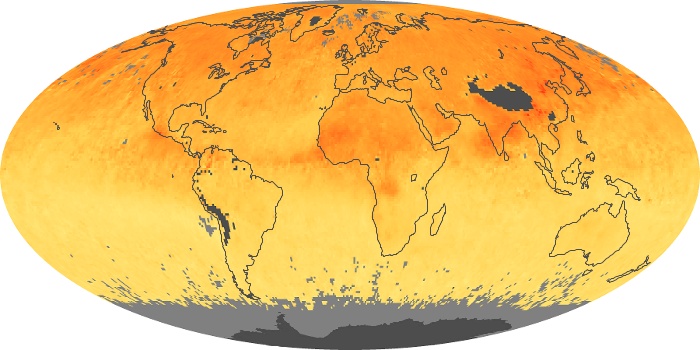
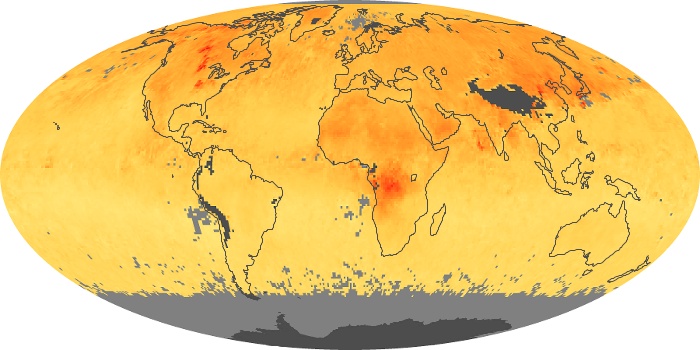
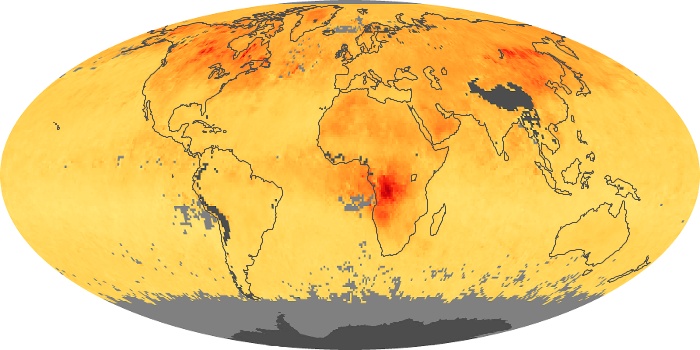
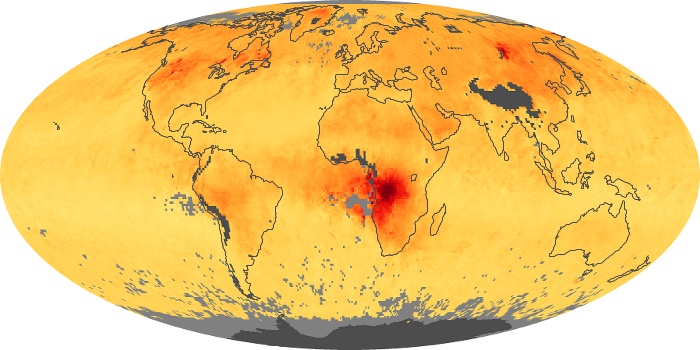
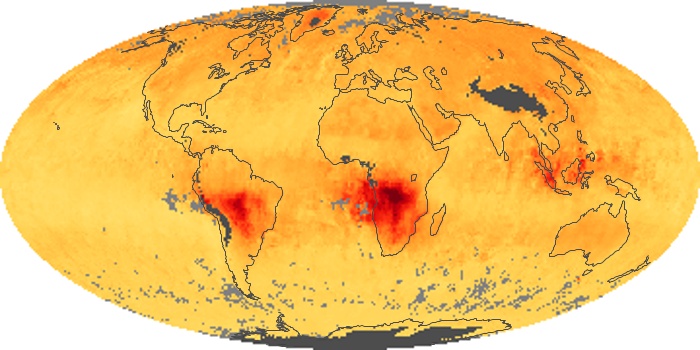
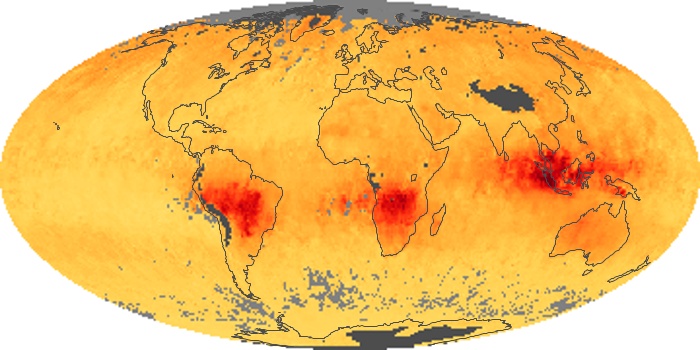
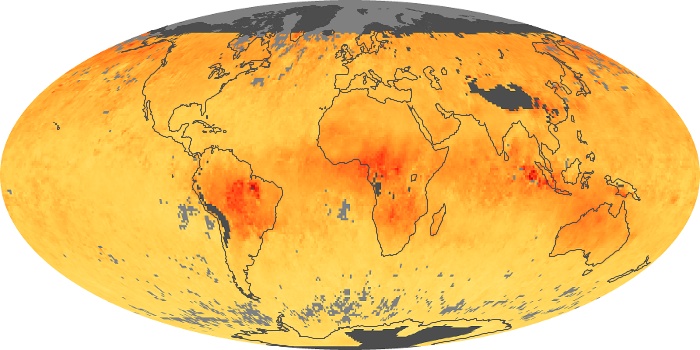
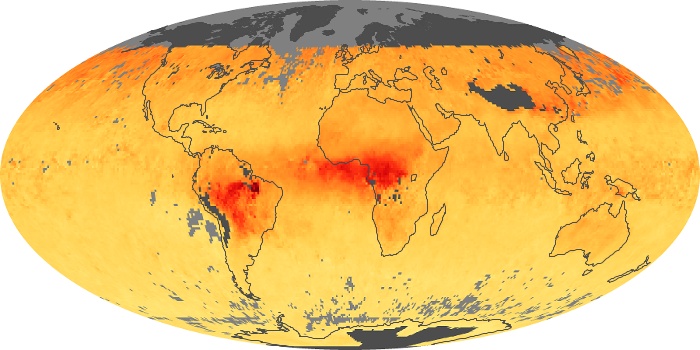
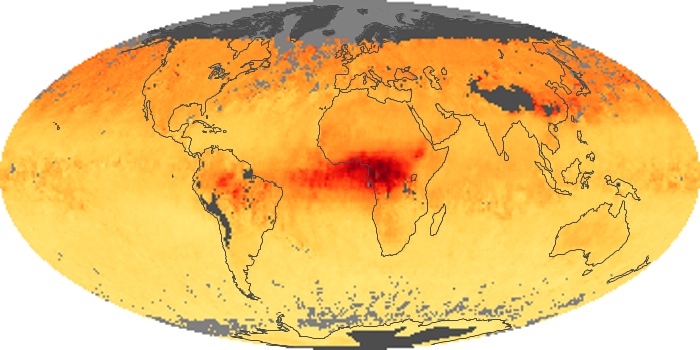
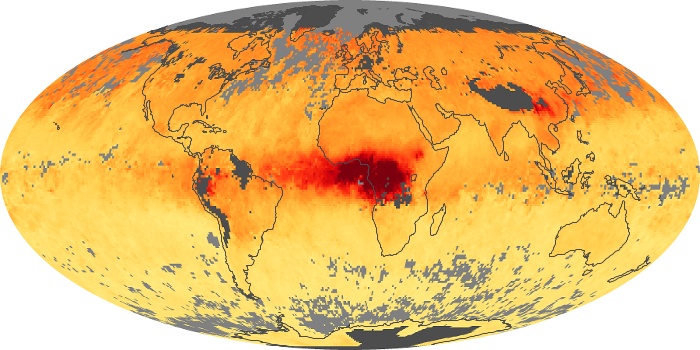
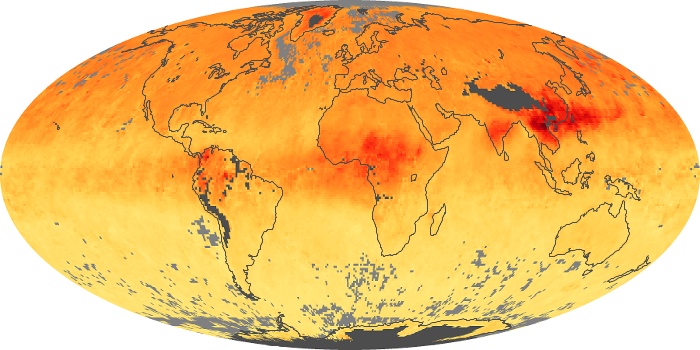
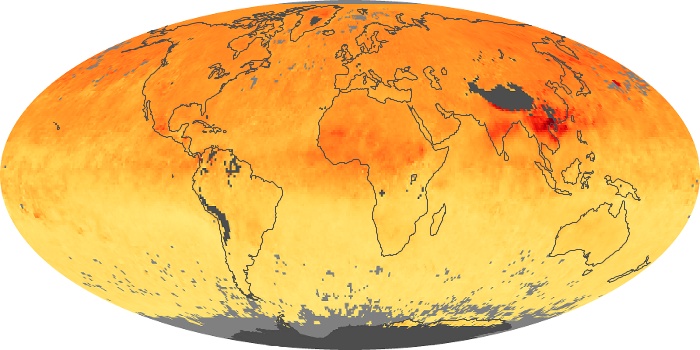
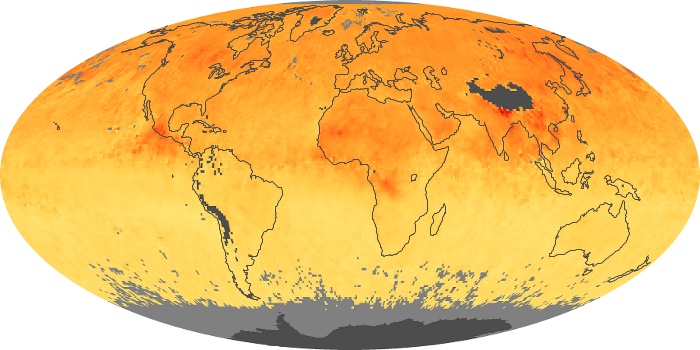
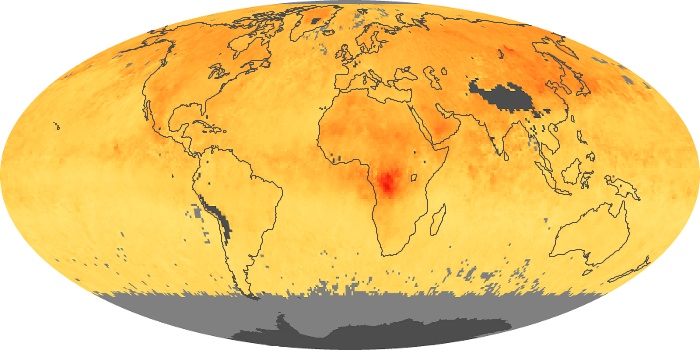
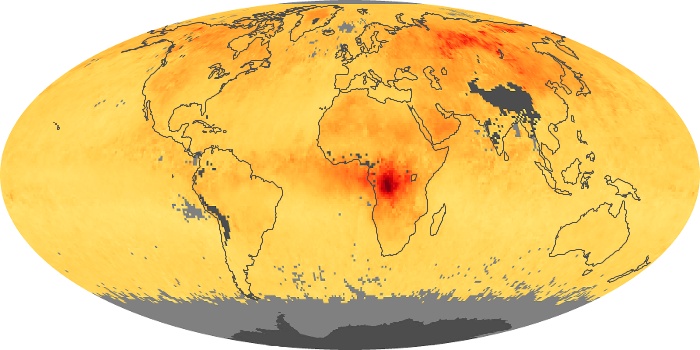
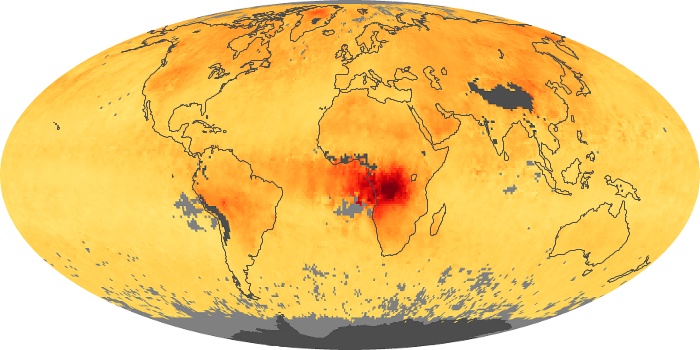
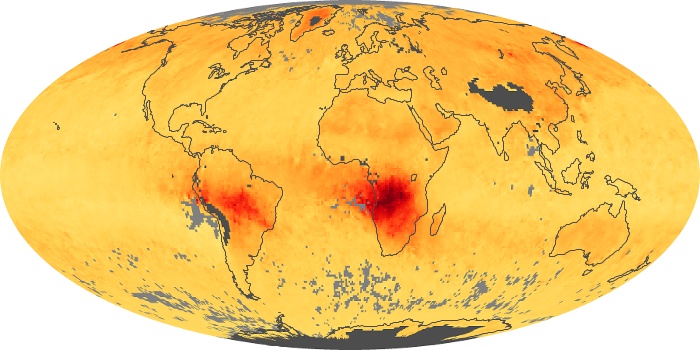
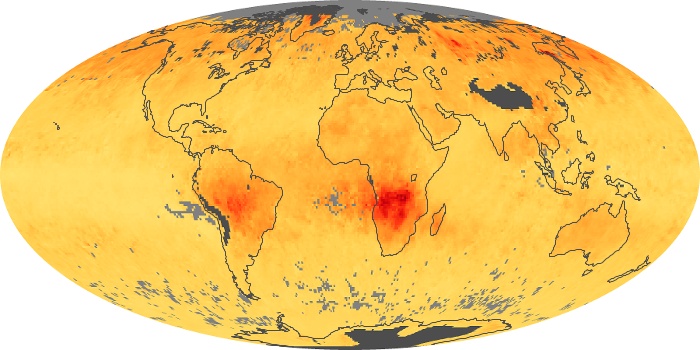
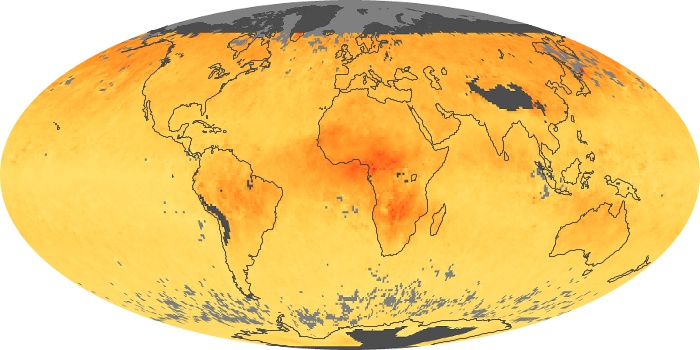
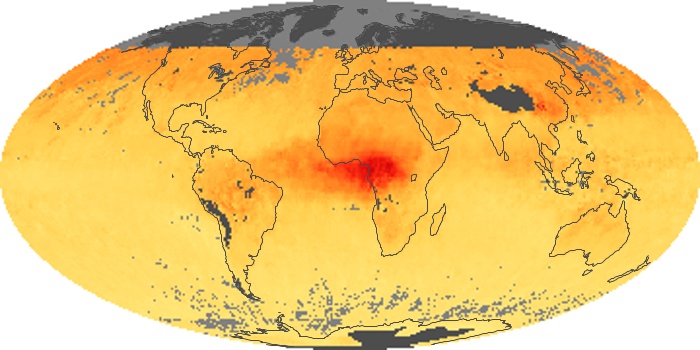
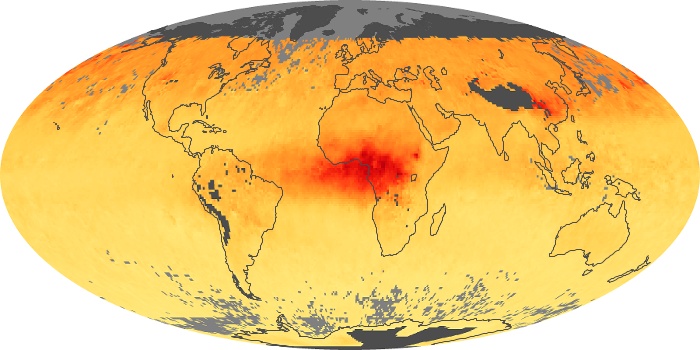
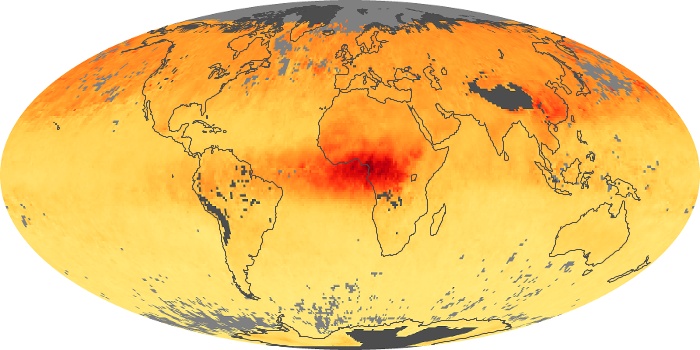
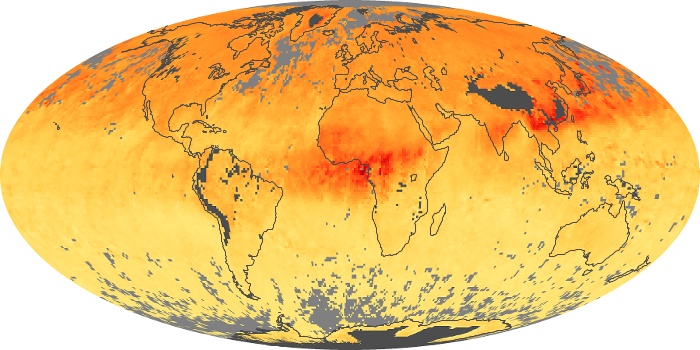
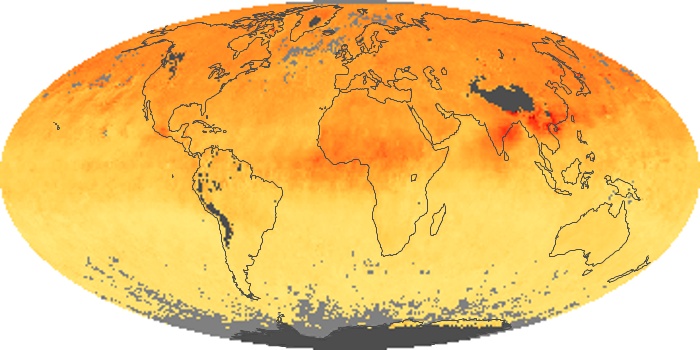
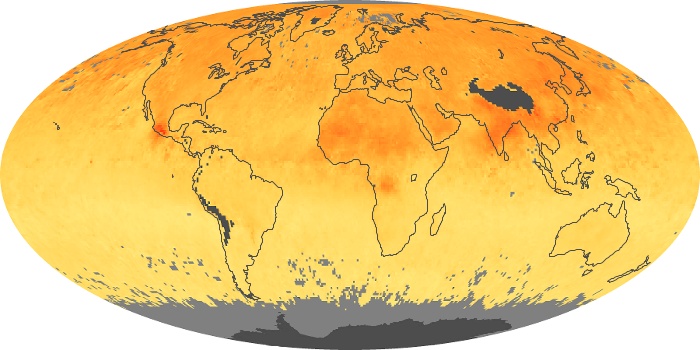
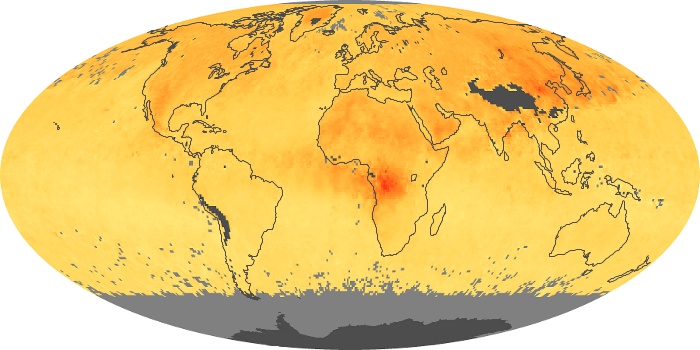
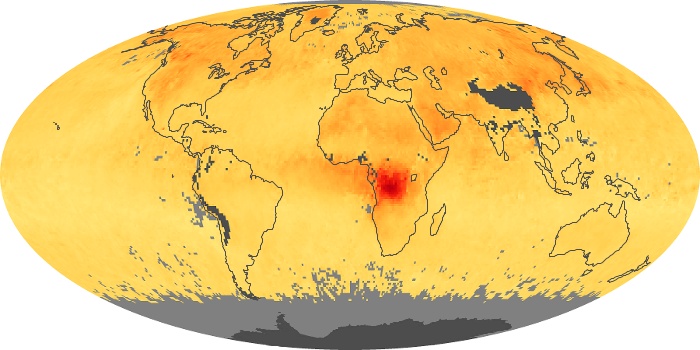
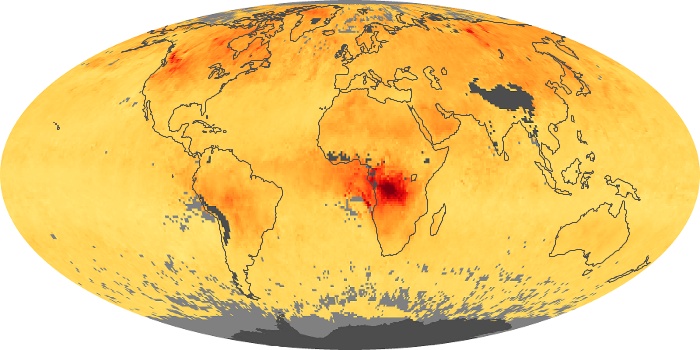
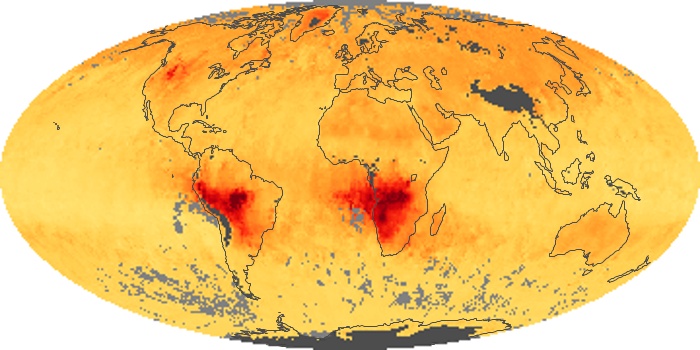
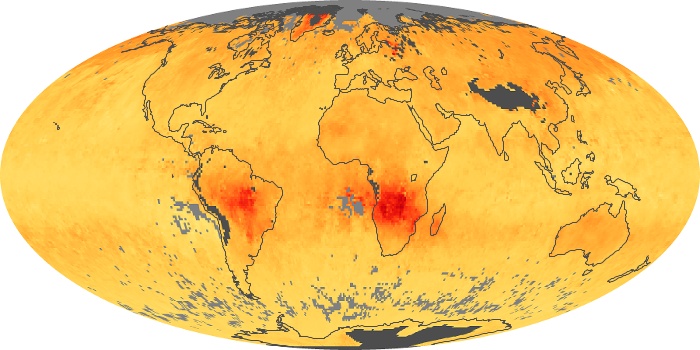
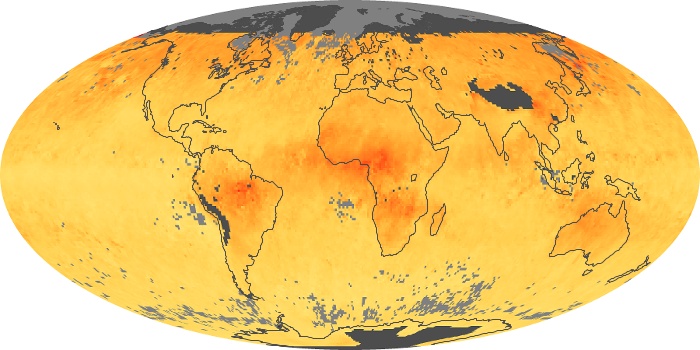
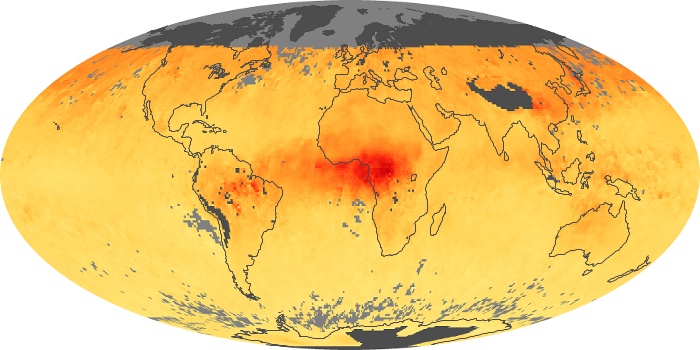
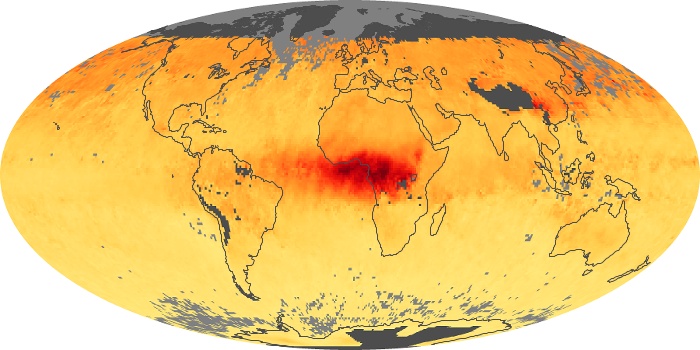
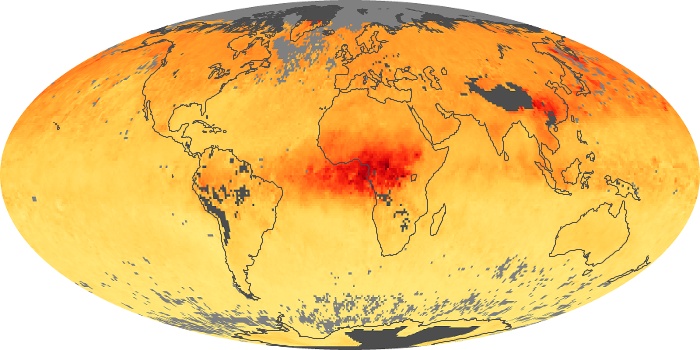
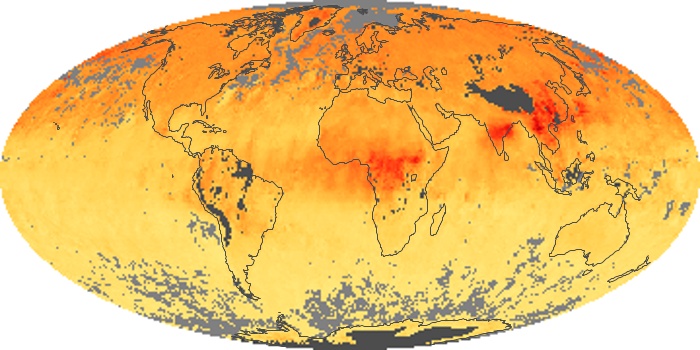
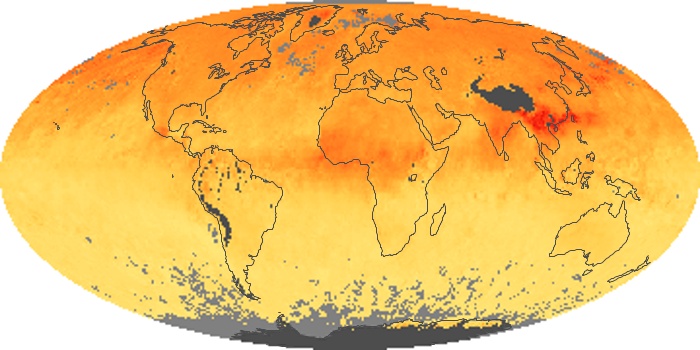
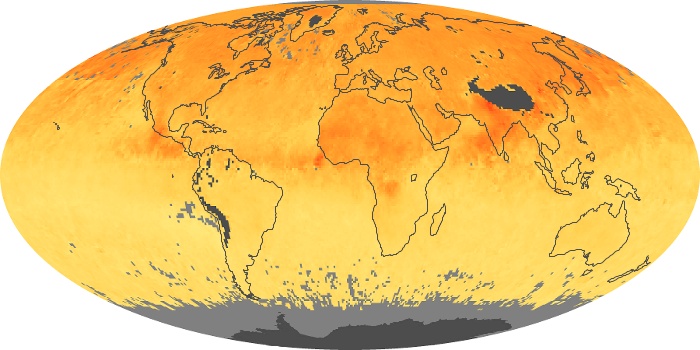
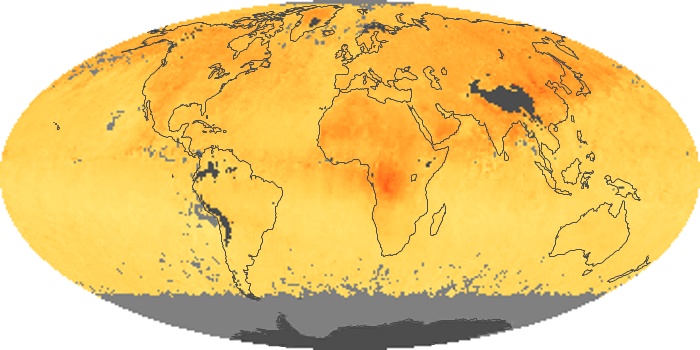
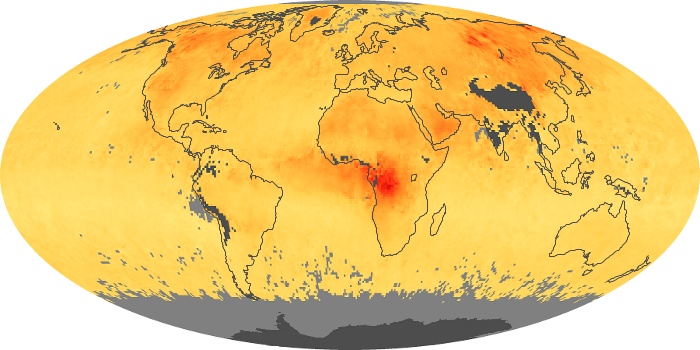
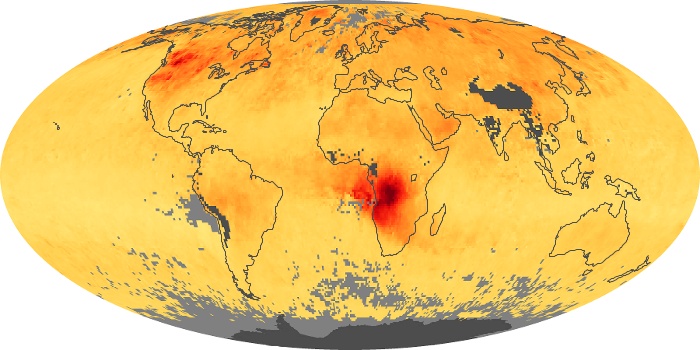
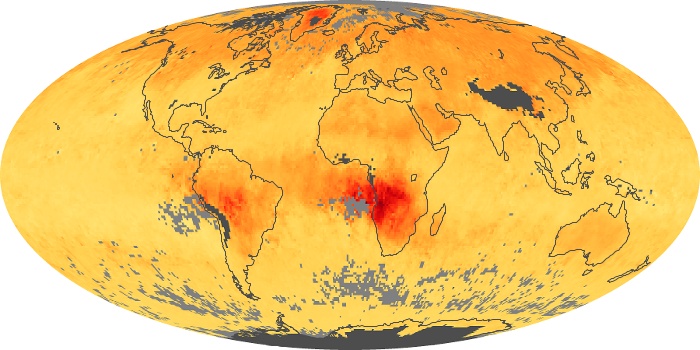
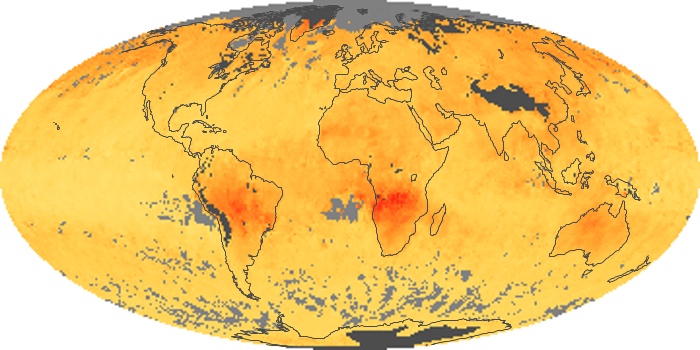
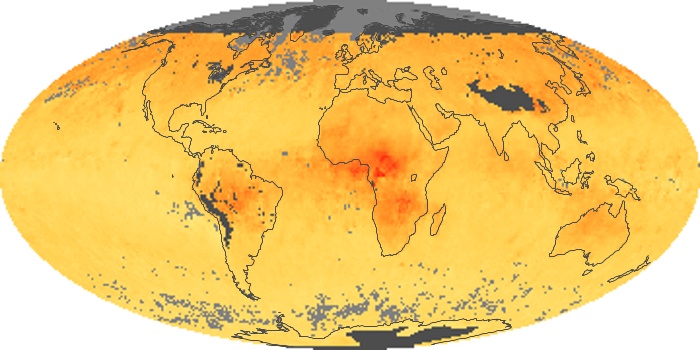
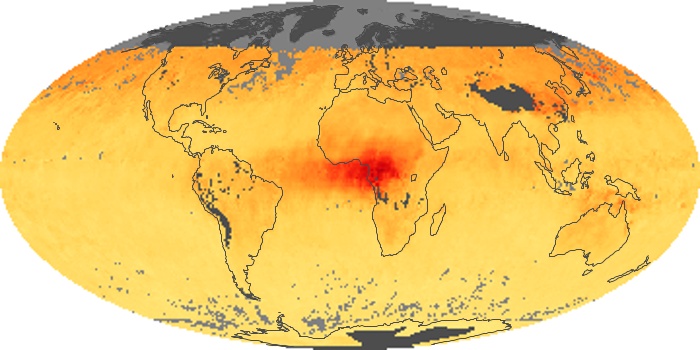
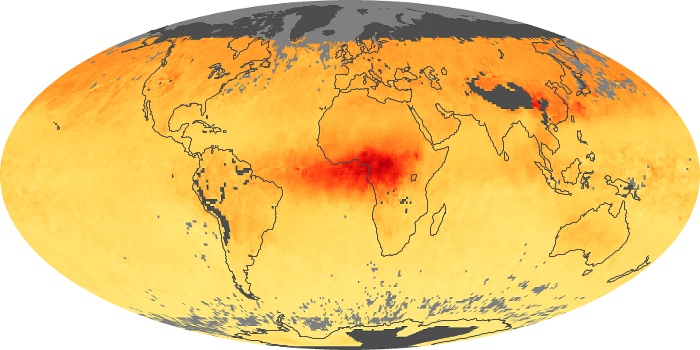
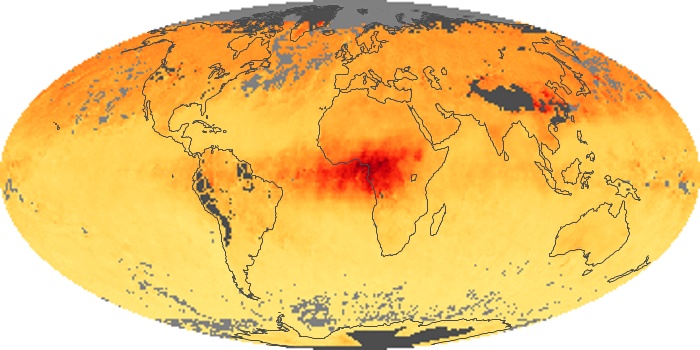
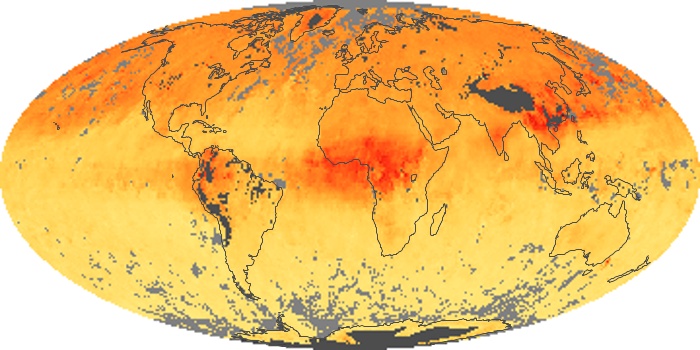
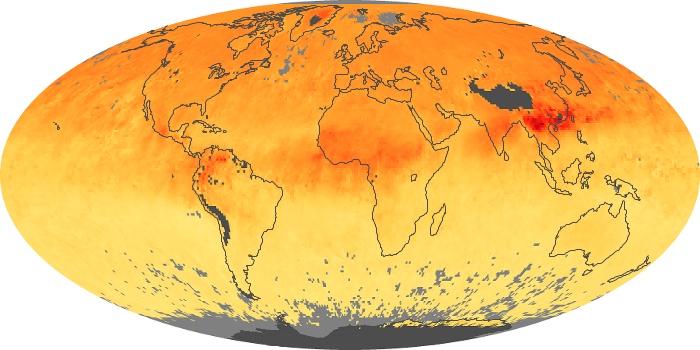
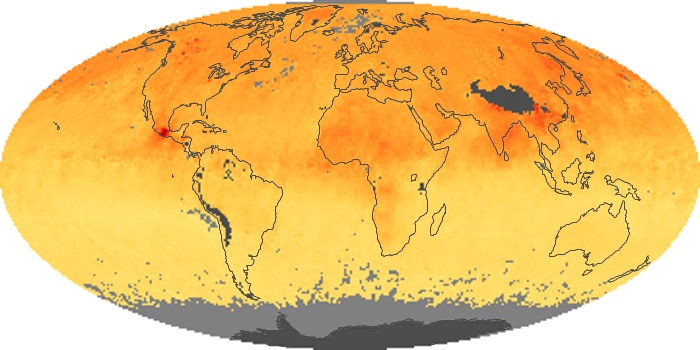
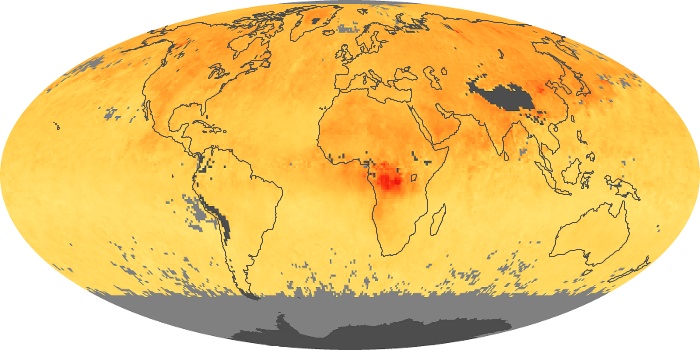
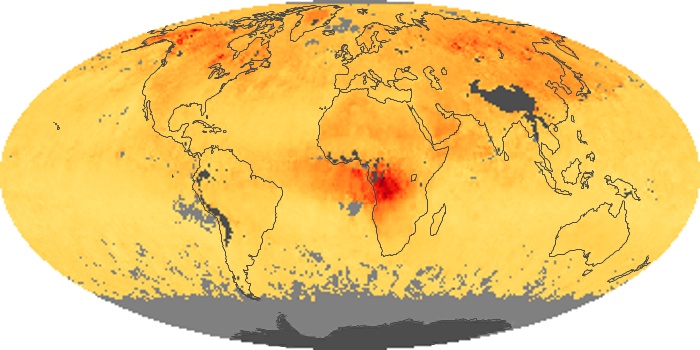
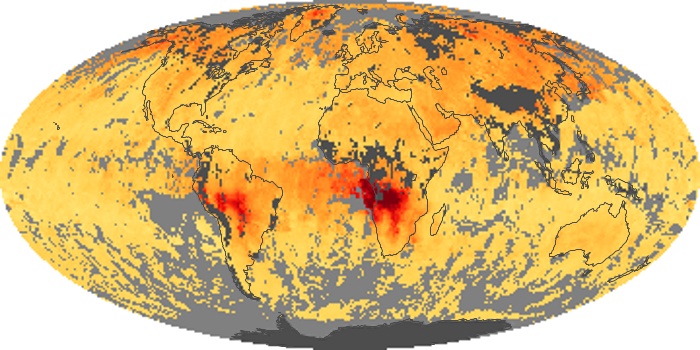
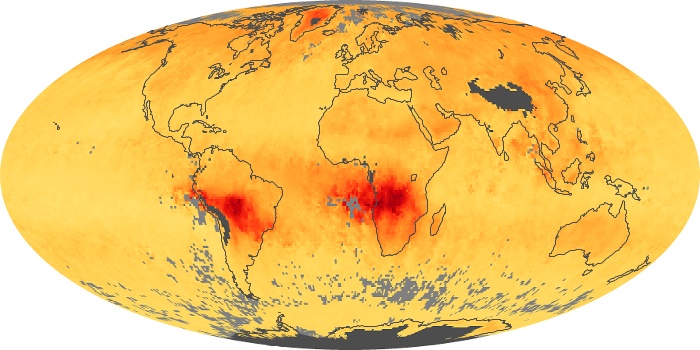
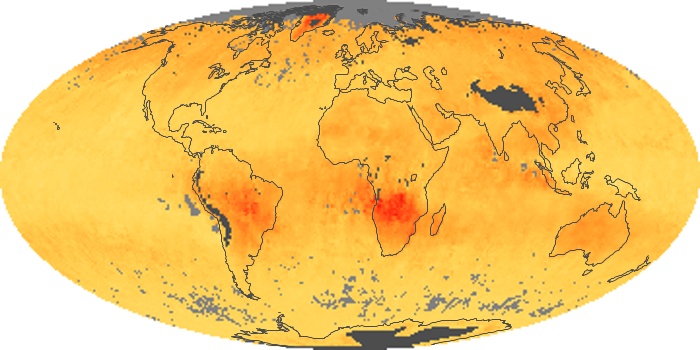
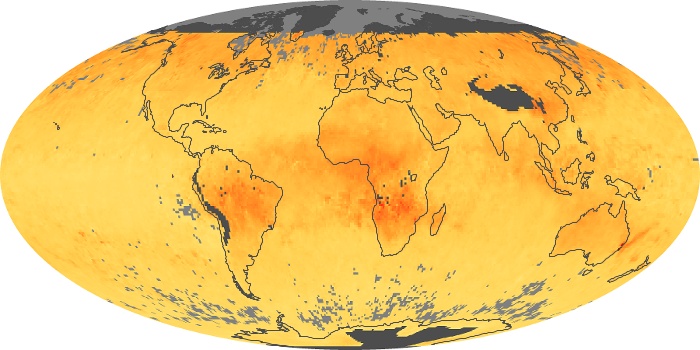
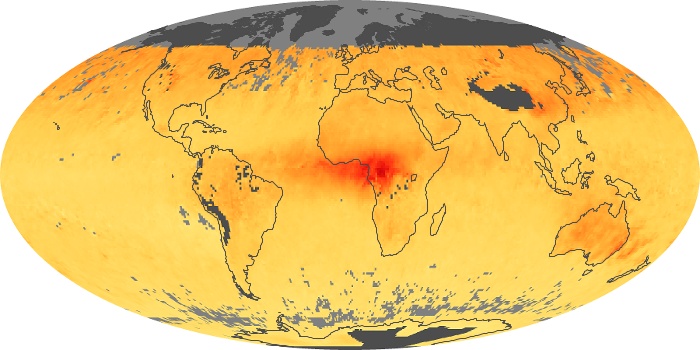
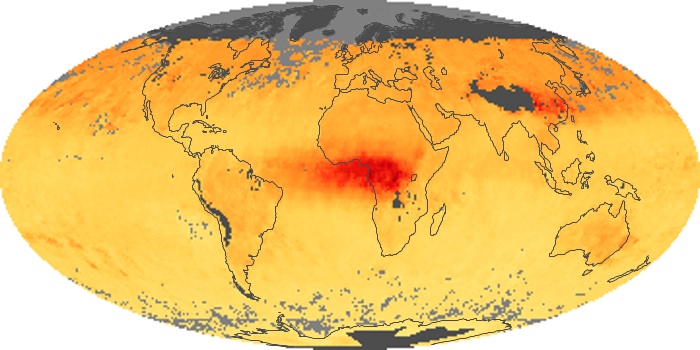
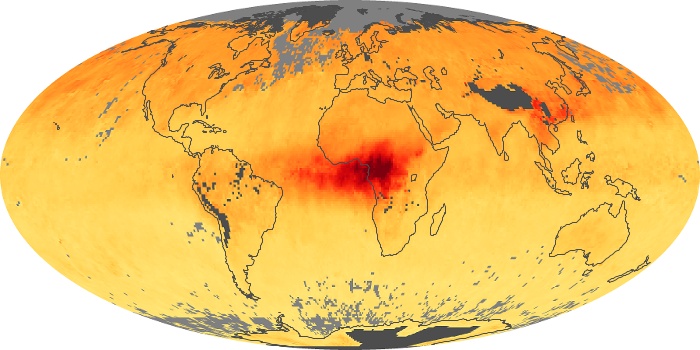
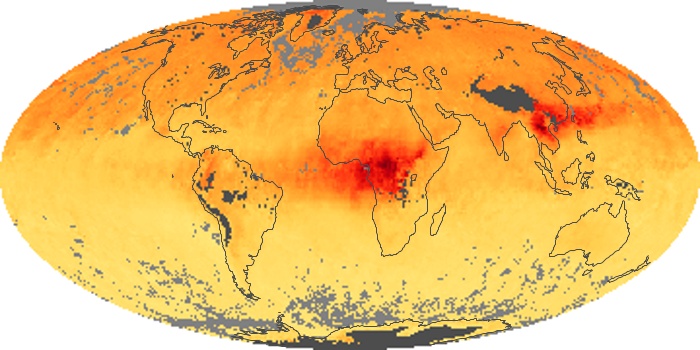
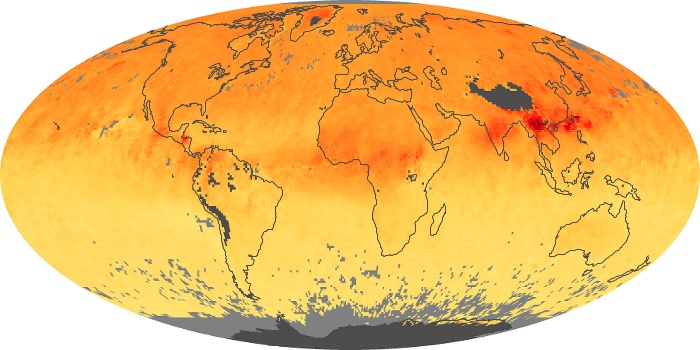

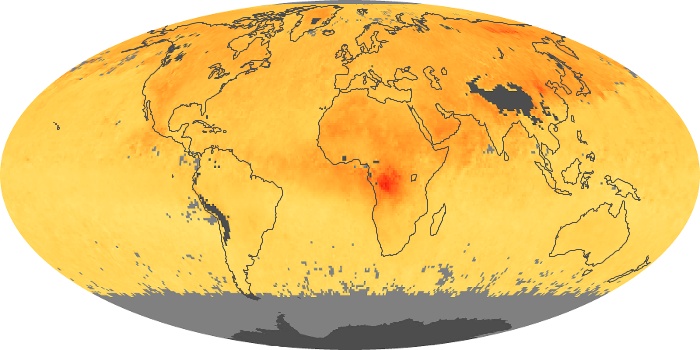
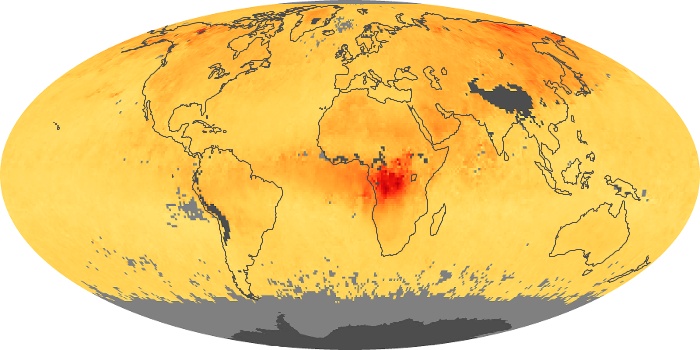
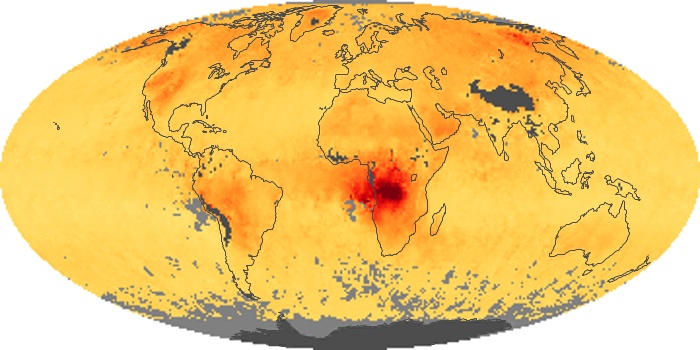
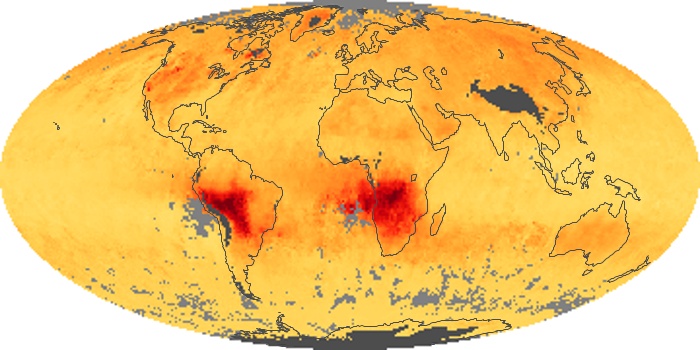
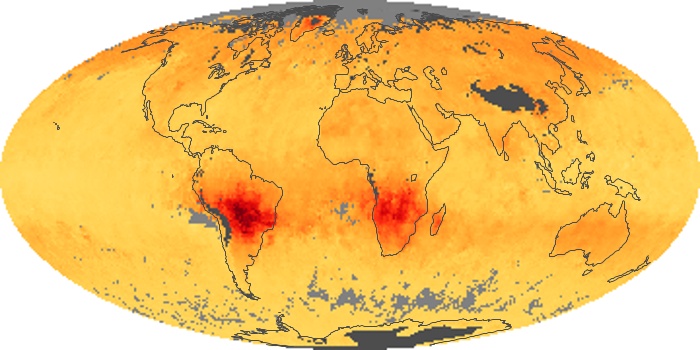
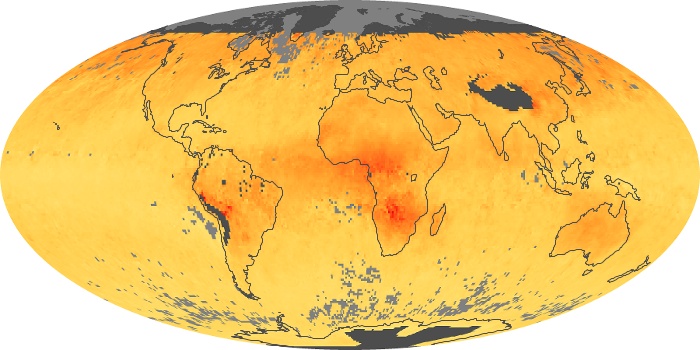
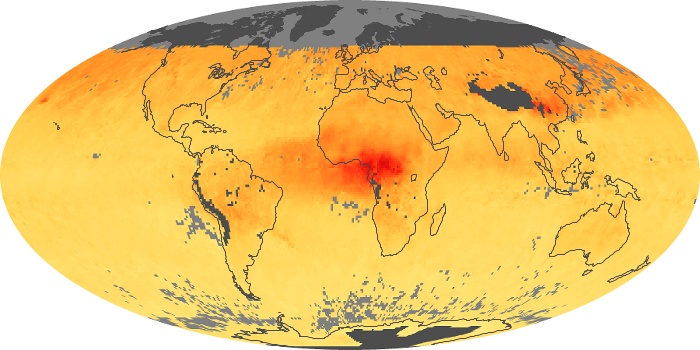
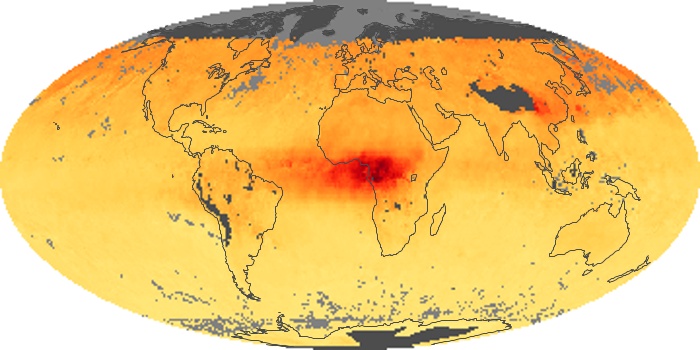
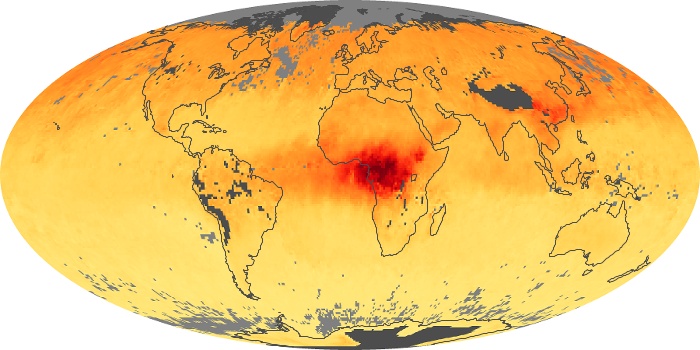
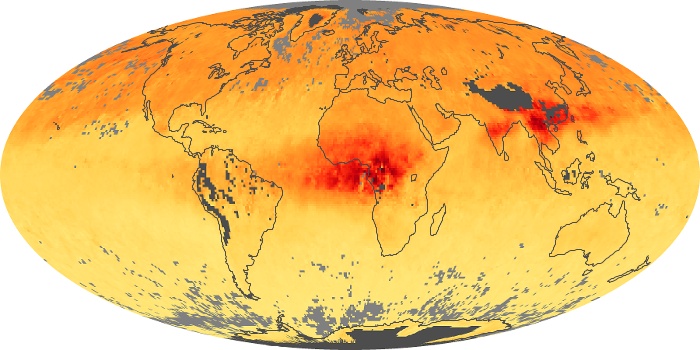
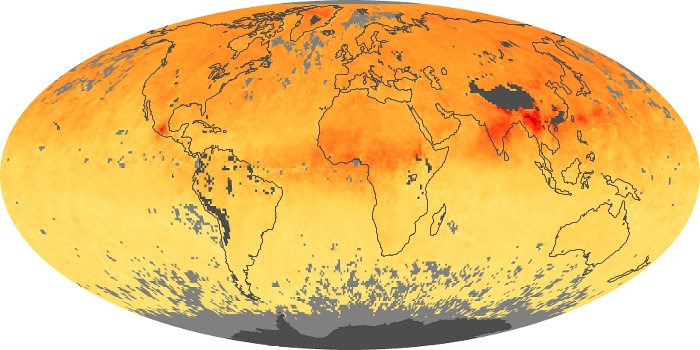
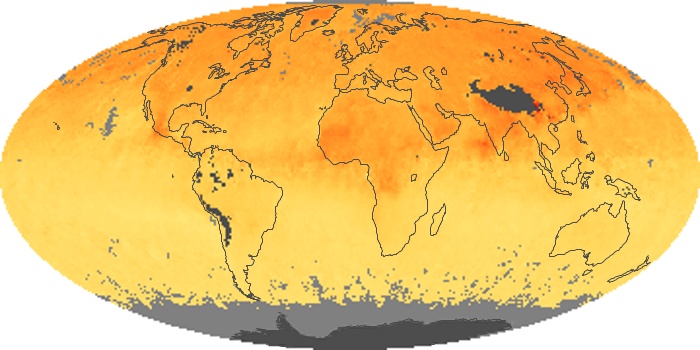
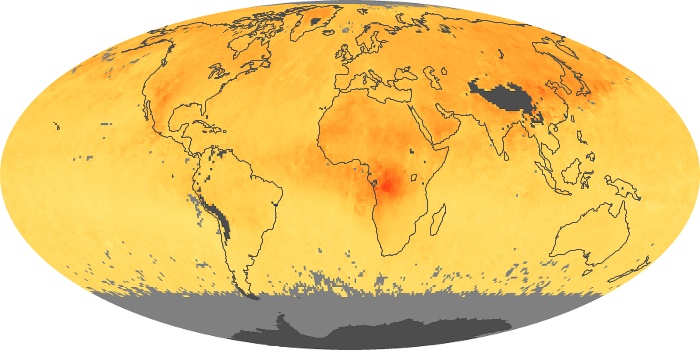
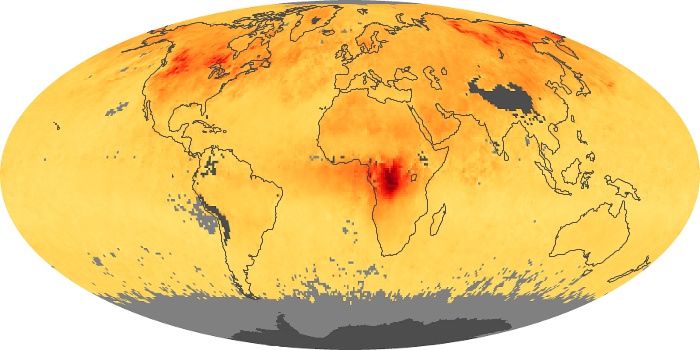
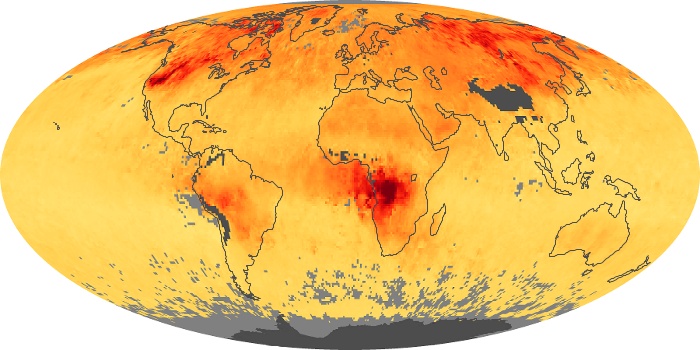
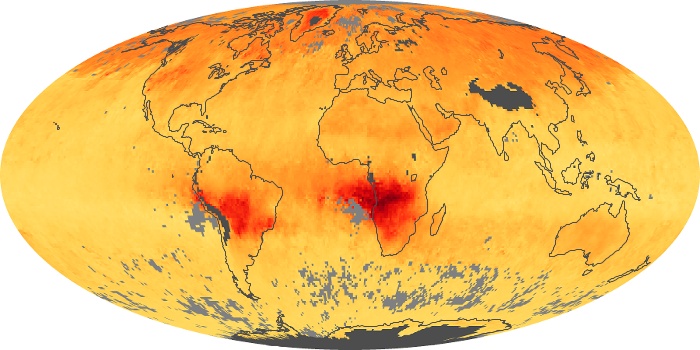
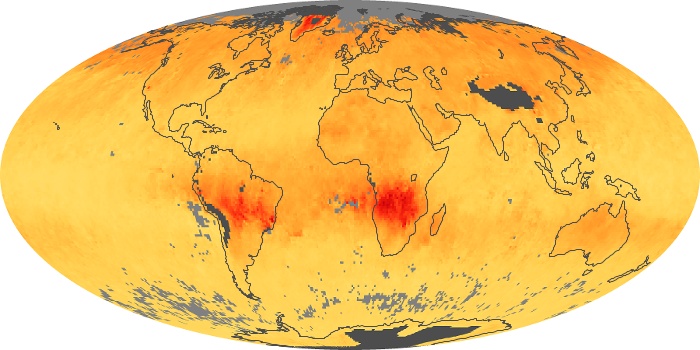
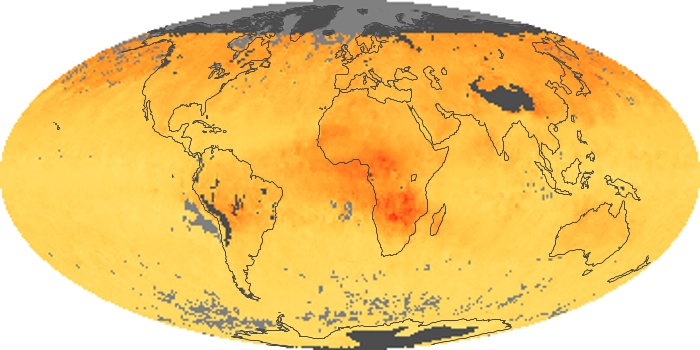
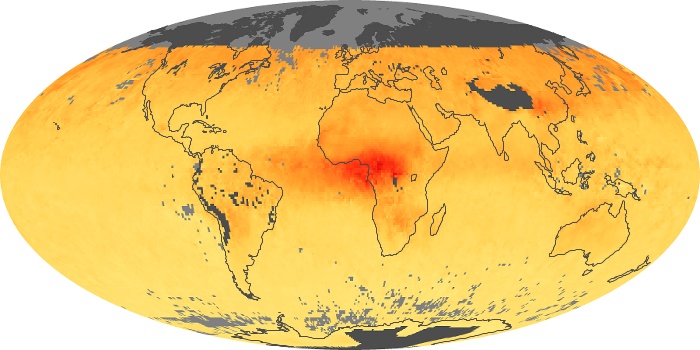
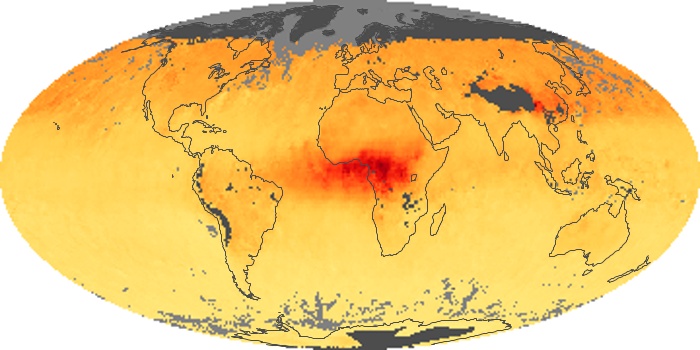
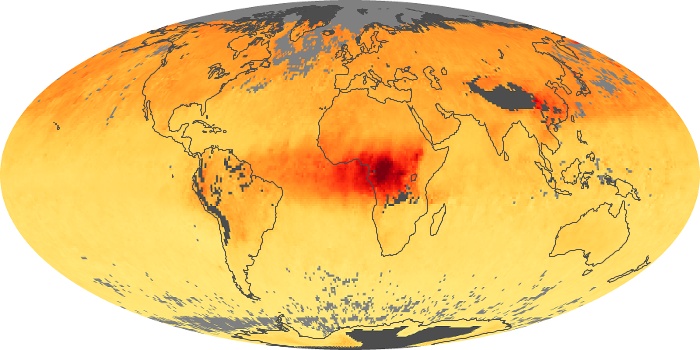
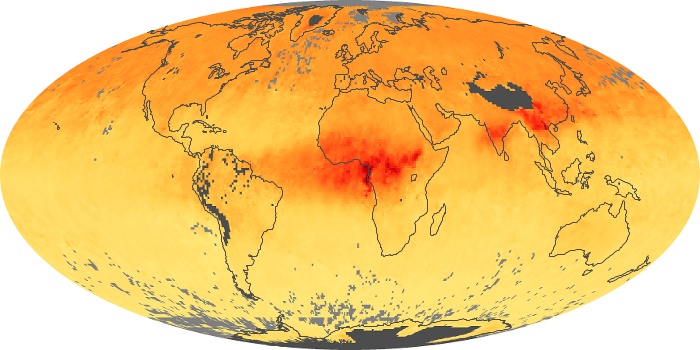
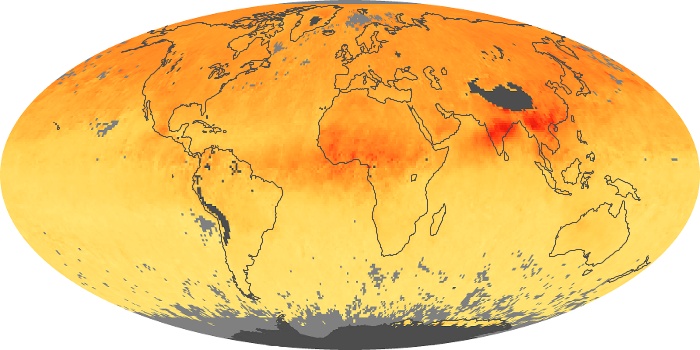
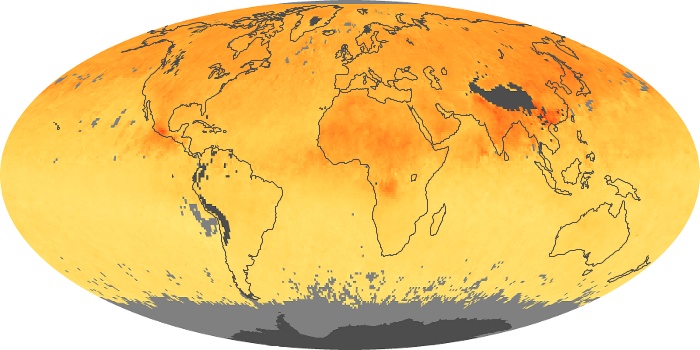
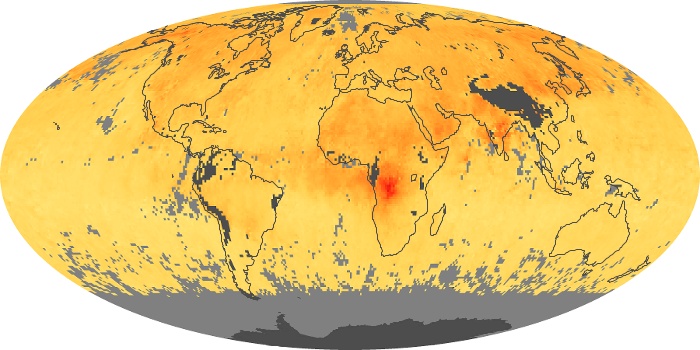
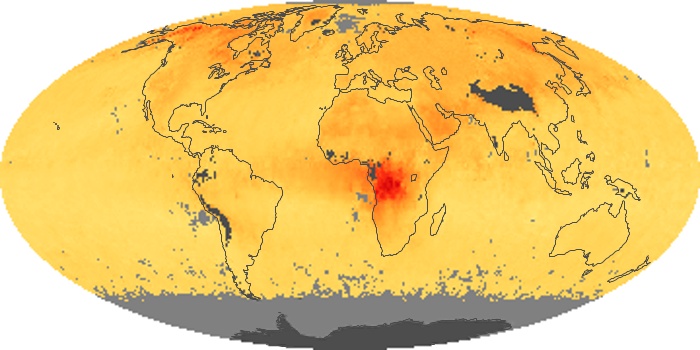
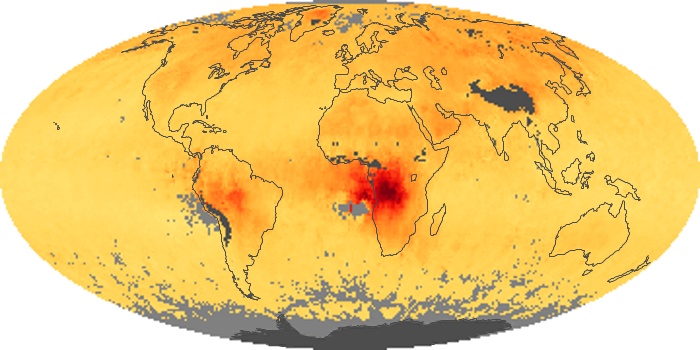
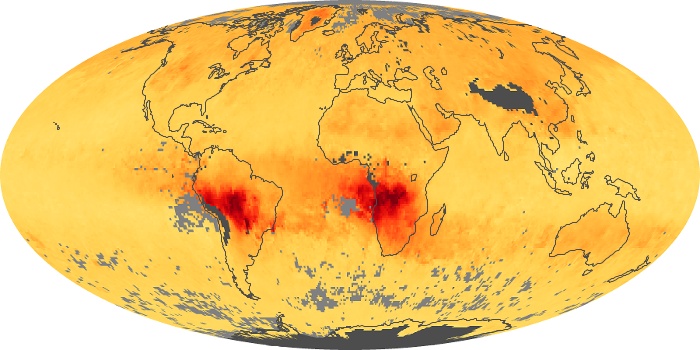

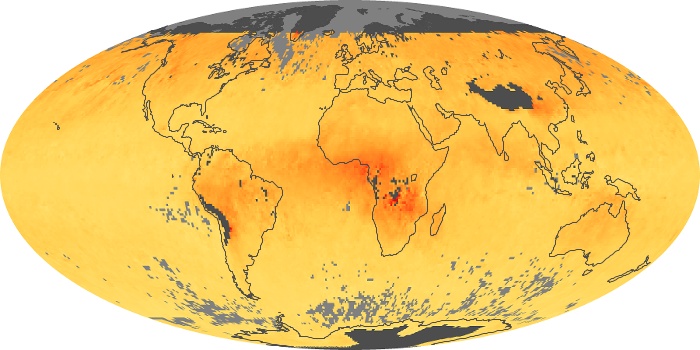
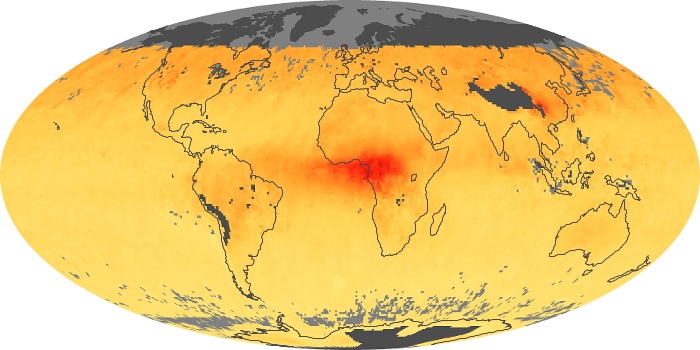
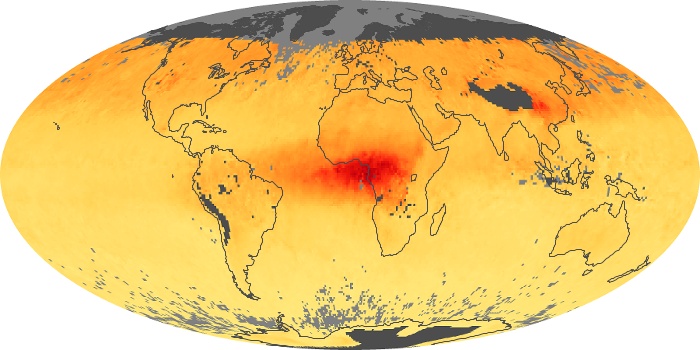
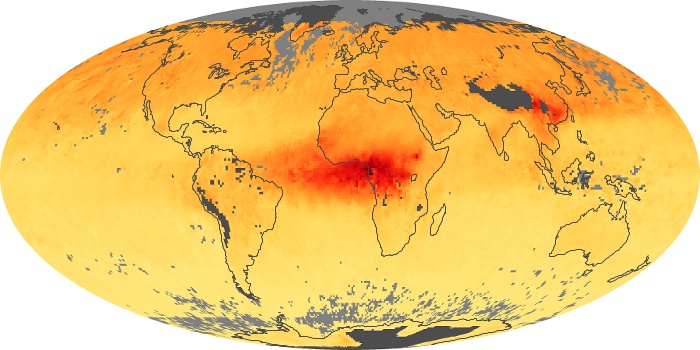
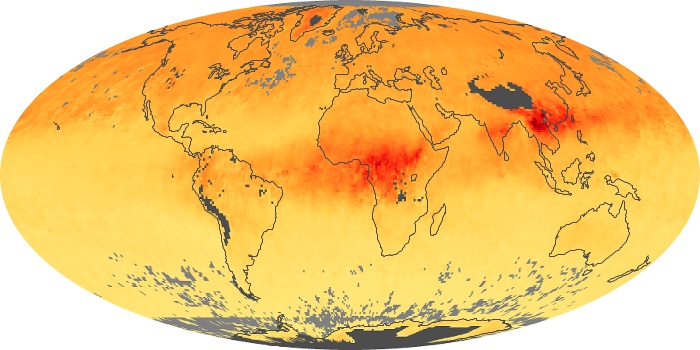
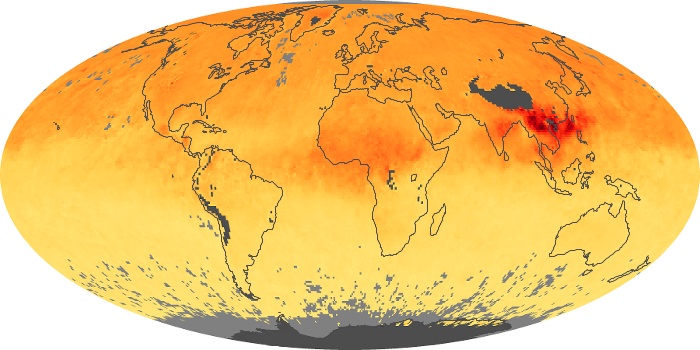
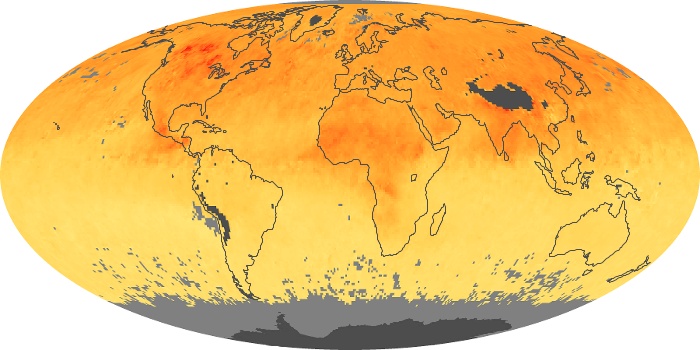
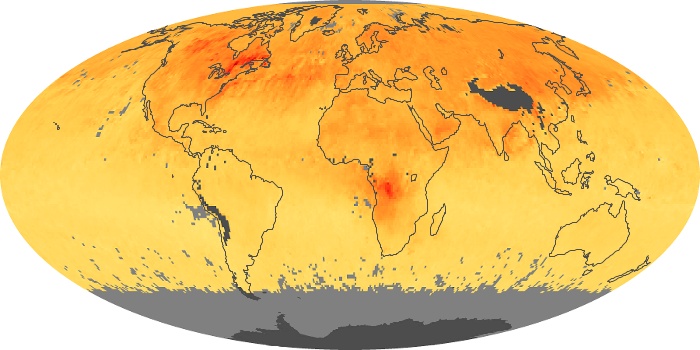
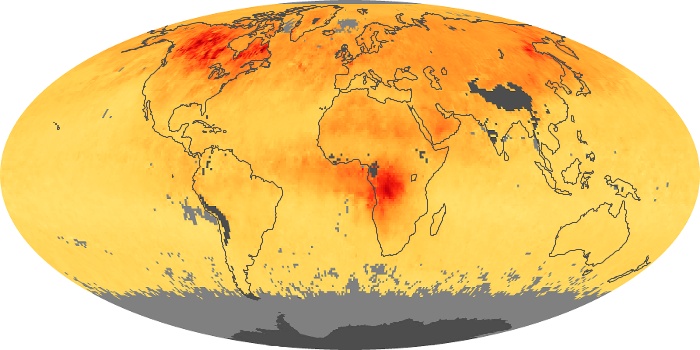
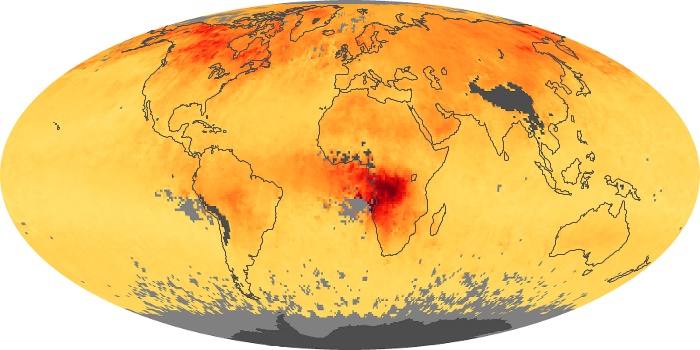
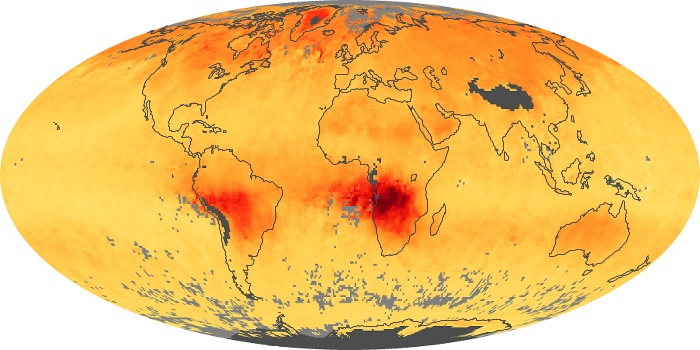
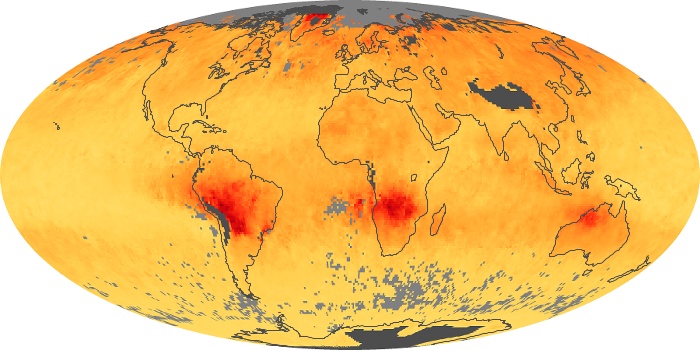
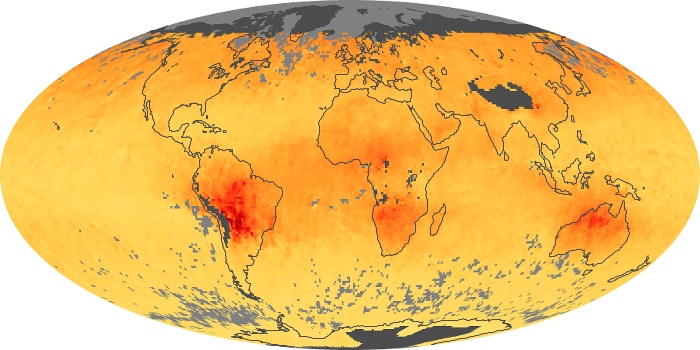
Colorless, odorless, and poisonous, carbon monoxide is one of the six major air pollutants regulated in the United States and in many other nations around the world. When carbon-based fuels, such as coal, wood, and oil, burn incompletely or inefficiently, they produce carbon monoxide. The gas is spread by winds and circulation patterns throughout the lower atmosphere (called the troposphere).
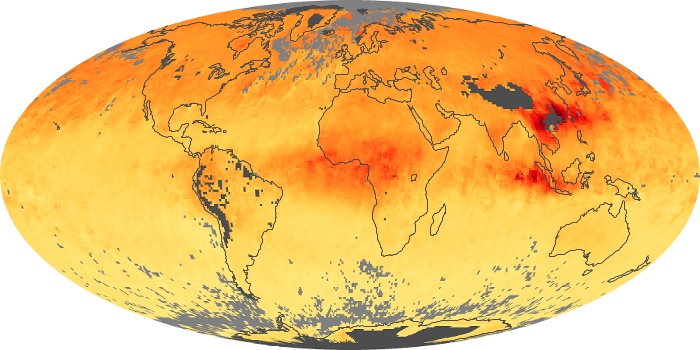
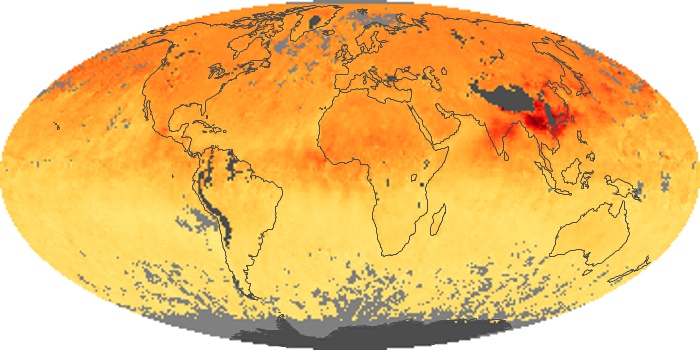
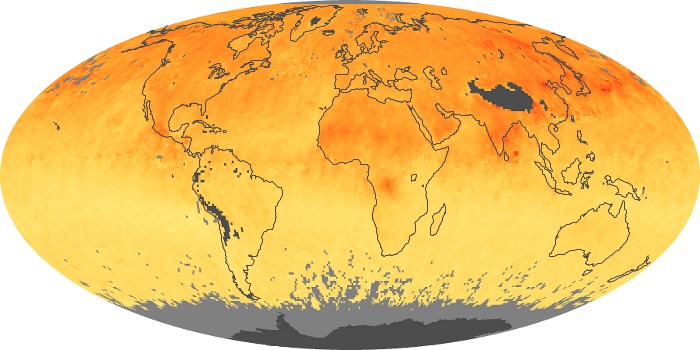
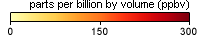
These maps show monthly averages of global concentrations of tropospheric carbon monoxide at an altitude of about 12,000 feet. The data were collected by the MOPITT (Measurements Of Pollution In The Troposphere) sensor on NASA’s Terra satellite. Concentrations of carbon monoxide are expressed in parts per billion by volume (ppbv). A concentration of 1 ppbv means that for every billion molecules of gas in the measured volume, one of them is a carbon monoxide molecule. Yellow areas have little or no carbon monoxide, while progressively higher concentrations are shown in orange and red. Places where the sensor didn’t collect data, perhaps due to clouds, are gray.
In different parts of the world and in different seasons, the amounts and sources of atmospheric carbon monoxide change. In Africa, for example, the seasonal shifts in carbon monoxide are tied to the widespread agricultural burning that shifts north and south of the equator with the seasons. Fires are an important source of carbon monoxide pollution in other regions of the Southern Hemisphere, such as the Amazon and Southeast Asia.
In the United States, Europe, and eastern China, on the other hand, the highest carbon monoxide concentrations occur around urban areas as a result of vehicle and industrial emissions. Fires burning over large areas in North America and Russia in some years can be an important source. The MOPITT observations often show that pollution emitted on one continent can travel across oceans to have a big impact on air quality on other continents.
Carbon monoxide is a trace gas in the atmosphere, and it does not have a direct effect on the global temperature, like methane and carbon dioxide do. However, carbon monoxide plays a major role in atmospheric chemistry, and it affects the ability of the atmosphere to cleanse itself of many other polluting gases. In combination with other pollutants and sunshine, it also takes part in the formation of lower-atmospheric (“bad”) ozone and urban smog.
View, download, or analyze more of these data from NASA Earth Observations (NEO):
Carbon Monoxide